QUESTION IMAGE
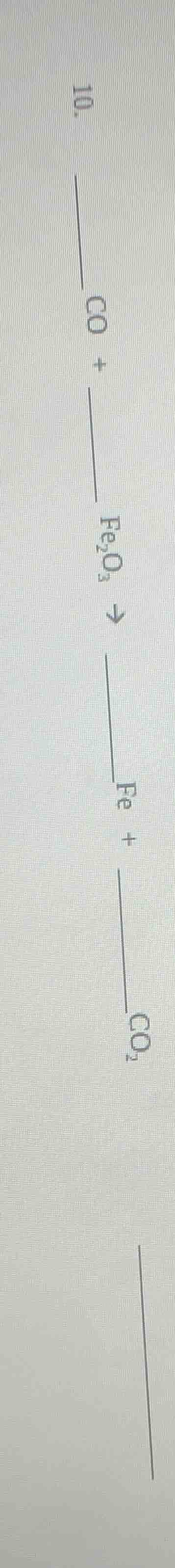
Question
Question was provided via image upload.
Step1: Balance Fe atoms
There are 2 Fe atoms on the left, so add coefficient 2 to Fe on the right:
$\text{CO} + \text{Fe}_2\text{O}_3
ightarrow 2\text{Fe} + \text{CO}_2$
Step2: Balance O atoms
Left side has 3 O from $\text{Fe}_2\text{O}_3$ + 1 O from $\text{CO}$; right side has 2 O from $\text{CO}_2$. To balance, add coefficient 3 to $\text{CO}$ and $\text{CO}_2$ (each $\text{CO}$ gains 1 O to become $\text{CO}_2$, 3 O atoms need 3 $\text{CO}$):
$3\text{CO} + \text{Fe}_2\text{O}_3
ightarrow 2\text{Fe} + 3\text{CO}_2$
Step3: Verify all atoms
Count atoms:
- C: 3 on left, 3 on right
- O: $3+3=6$ on left, $3\times2=6$ on right
- Fe: 2 on left, 2 on right
All atoms are balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$3\text{CO} + \text{Fe}_2\text{O}_3
ightarrow 2\text{Fe} + 3\text{CO}_2$
(The coefficients in order are 3, 1, 2, 3)