QUESTION IMAGE
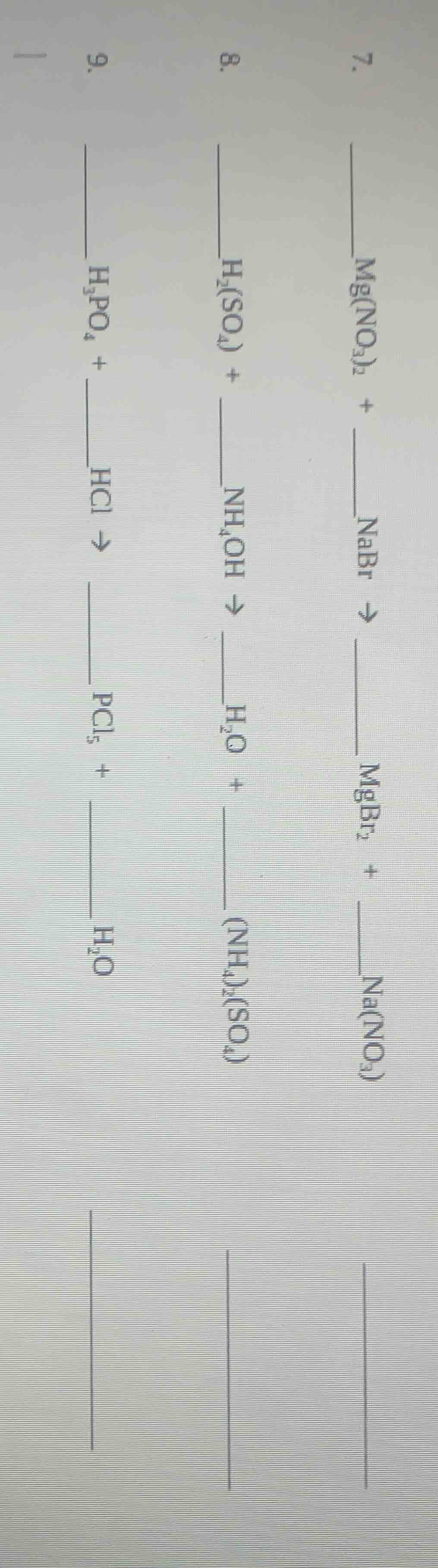
Question
Question was provided via image upload.
Step1: Balance equation 7 (double displacement)
First, match cations/anions: $\text{Mg(NO}_3\text{)}_2 + 2\text{NaBr}
ightarrow \text{MgBr}_2 + 2\text{NaNO}_3$
Count atoms: Mg:1=1, N:2=2, O:6=6, Na:2=2, Br:2=2. Balanced.
Step2: Balance equation 8 (acid-base neutralization)
Match ions, balance $\text{NH}_4^+$ and $\text{OH}^-$: $\text{H}_2\text{SO}_4 + 2\text{NH}_4\text{OH}
ightarrow 2\text{H}_2\text{O} + (\text{NH}_4)_2\text{SO}_4$
Count atoms: H:2+8=10; 4+8=12? Correct: $\text{H}_2\text{SO}_4$ has 2 H, $2\text{NH}_4\text{OH}$ has 2*(4+1)=10 H → total 12 H. $2\text{H}_2\text{O}$ has 4 H, $(\text{NH}_4)_2\text{SO}_4$ has 8 H → 12 H. S:1=1, O:4+2=6; 2+4=6. N:2=2. Balanced.
Step3: Balance equation 9 (double displacement)
Rearrange reactants/products, balance Cl and P: $\text{H}_3\text{PO}_4 + 5\text{HCl}
ightarrow \text{PCl}_5 + 4\text{H}_2\text{O}$
Count atoms: H:3+5=8; 8=8. P:1=1. O:4=4. Cl:5=5. Balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\boldsymbol{1\text{Mg(NO}_3\text{)}_2 + 2\text{NaBr}
ightarrow 1\text{MgBr}_2 + 2\text{NaNO}_3}$
- $\boldsymbol{1\text{H}_2\text{SO}_4 + 2\text{NH}_4\text{OH}
ightarrow 2\text{H}_2\text{O} + 1(\text{NH}_4)_2\text{SO}_4}$
- $\boldsymbol{1\text{H}_3\text{PO}_4 + 5\text{HCl}
ightarrow 1\text{PCl}_5 + 4\text{H}_2\text{O}}$
(Note: The coefficients 1 are optional but included for clarity; omitting them is also acceptable in balanced equations.)