QUESTION IMAGE
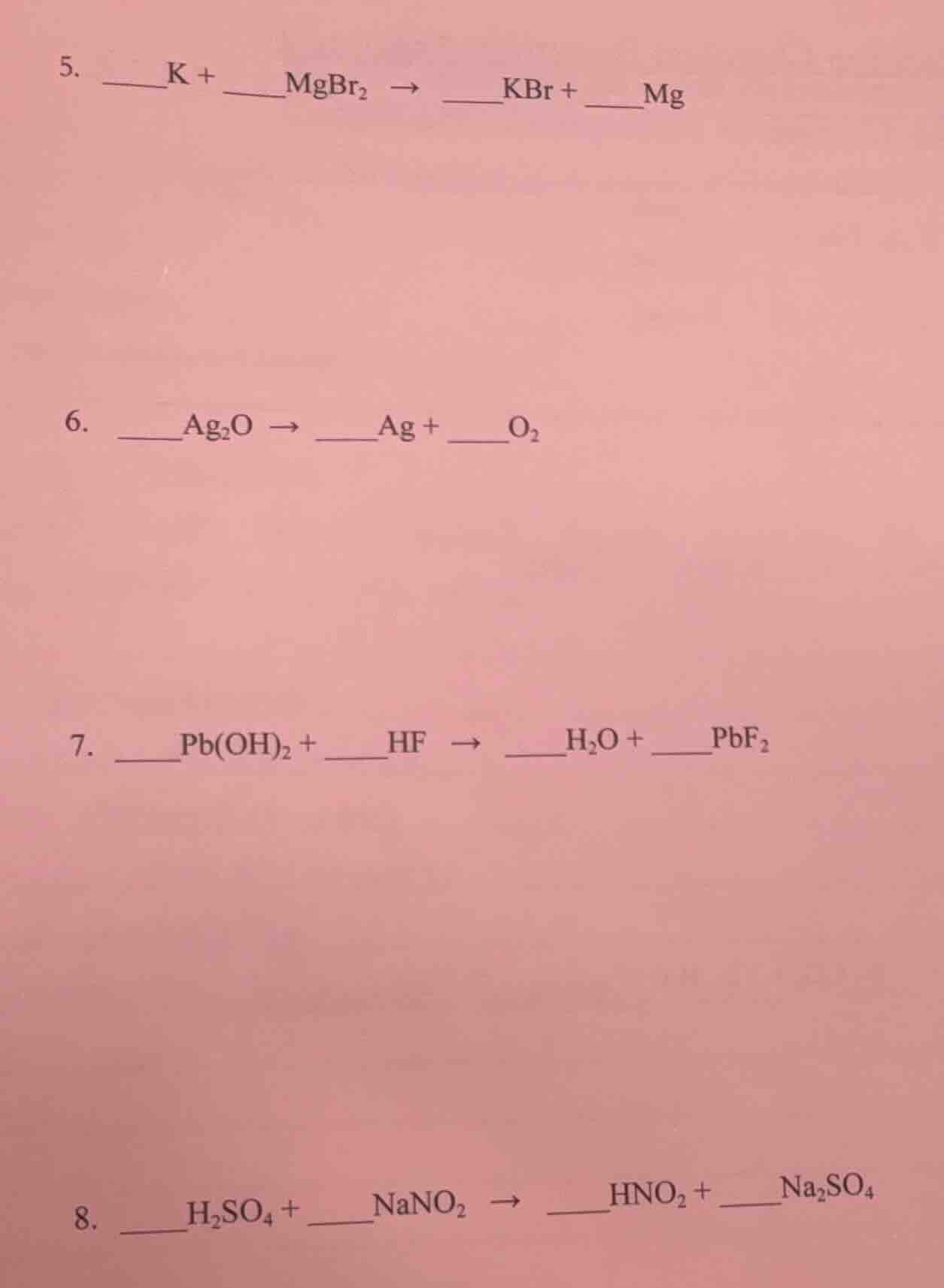
Question
Question was provided via image upload.
Step1: Balance Br atoms
On reactant side: 2 Br in $\text{MgBr}_2$; product side: 1 Br in $\text{KBr}$. Add coefficient 2 to $\text{KBr}$:
$\underline{\quad}\text{K} + \text{MgBr}_2
ightarrow 2\text{KBr} + \underline{\quad}\text{Mg}$
Step2: Balance K atoms
Product side: 2 K in $2\text{KBr}$; reactant side: 1 K. Add coefficient 2 to $\text{K}$:
$2\text{K} + \text{MgBr}_2
ightarrow 2\text{KBr} + \underline{\quad}\text{Mg}$
Step3: Balance Mg atoms
Reactant side: 1 Mg in $\text{MgBr}_2$; product side: 1 Mg. Add coefficient 1 to $\text{Mg}$:
$2\text{K} + \text{MgBr}_2
ightarrow 2\text{KBr} + \text{Mg}$
---
Step1: Balance O atoms
Reactant side: 1 O in $\text{Ag}_2\text{O}$; product side: 2 O in $\text{O}_2$. Add coefficient 2 to $\text{Ag}_2\text{O}$:
$2\text{Ag}_2\text{O}
ightarrow \underline{\quad}\text{Ag} + \text{O}_2$
Step2: Balance Ag atoms
Reactant side: 4 Ag in $2\text{Ag}_2\text{O}$; product side: 1 Ag. Add coefficient 4 to $\text{Ag}$:
$2\text{Ag}_2\text{O}
ightarrow 4\text{Ag} + \text{O}_2$
---
Step1: Balance F atoms
Reactant side: 1 F in $\text{HF}$; product side: 2 F in $\text{PbF}_2$. Add coefficient 2 to $\text{HF}$:
$\text{Pb(OH)}_2 + 2\text{HF}
ightarrow \underline{\quad}\text{H}_2\text{O} + \text{PbF}_2$
Step2: Balance H and O atoms
Reactant side: 4 H ($2$ from $\text{Pb(OH)}_2$, $2$ from $2\text{HF}$) and 2 O; product side: 2 H in $\text{H}_2\text{O}$. Add coefficient 2 to $\text{H}_2\text{O}$:
$\text{Pb(OH)}_2 + 2\text{HF}
ightarrow 2\text{H}_2\text{O} + \text{PbF}_2$
---
Step1: Balance Na atoms
Reactant side: 1 Na in $\text{NaNO}_2$; product side: 2 Na in $\text{Na}_2\text{SO}_4$. Add coefficient 2 to $\text{NaNO}_2$:
$\underline{\quad}\text{H}_2\text{SO}_4 + 2\text{NaNO}_2
ightarrow \underline{\quad}\text{HNO}_2 + \text{Na}_2\text{SO}_4$
Step2: Balance N atoms
Reactant side: 2 N in $2\text{NaNO}_2$; product side: 1 N in $\text{HNO}_2$. Add coefficient 2 to $\text{HNO}_2$:
$\underline{\quad}\text{H}_2\text{SO}_4 + 2\text{NaNO}_2
ightarrow 2\text{HNO}_2 + \text{Na}_2\text{SO}_4$
Step3: Balance S, H, O atoms
All remaining atoms are balanced with coefficient 1 for $\text{H}_2\text{SO}_4$:
$\text{H}_2\text{SO}_4 + 2\text{NaNO}_2
ightarrow 2\text{HNO}_2 + \text{Na}_2\text{SO}_4$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $2\text{K} + \text{MgBr}_2
ightarrow 2\text{KBr} + \text{Mg}$
- $2\text{Ag}_2\text{O}
ightarrow 4\text{Ag} + \text{O}_2$
- $\text{Pb(OH)}_2 + 2\text{HF}
ightarrow 2\text{H}_2\text{O} + \text{PbF}_2$
- $\text{H}_2\text{SO}_4 + 2\text{NaNO}_2
ightarrow 2\text{HNO}_2 + \text{Na}_2\text{SO}_4$