QUESTION IMAGE
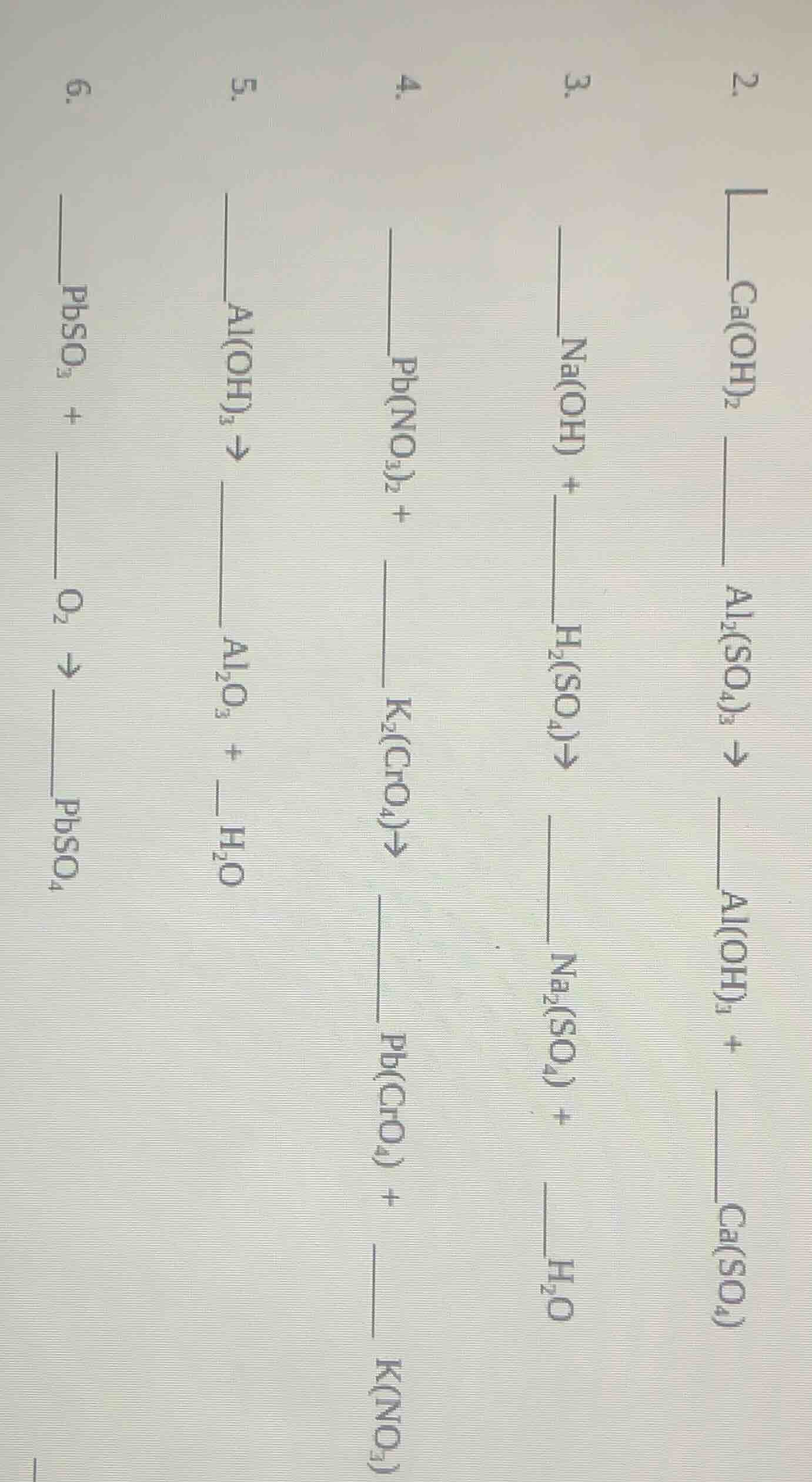
Question
Question was provided via image upload.
Step1: Balance Q2: Match Al atoms
$$3\text{Ca(OH)}_2 + \text{Al}_2(\text{SO}_4)_3
ightarrow 2\text{Al(OH)}_3 + \text{Ca(SO}_4)$$
Step2: Balance Q2: Match Ca/SO₄ atoms
$$3\text{Ca(OH)}_2 + \text{Al}_2(\text{SO}_4)_3
ightarrow 2\text{Al(OH)}_3 + 3\text{Ca(SO}_4)$$
Step3: Balance Q3: Match Na atoms
$$2\text{NaOH} + \text{H}_2(\text{SO}_4)
ightarrow \text{Na}_2(\text{SO}_4) + \text{H}_2\text{O}$$
Step4: Balance Q3: Match H/O atoms
$$2\text{NaOH} + \text{H}_2(\text{SO}_4)
ightarrow \text{Na}_2(\text{SO}_4) + 2\text{H}_2\text{O}$$
Step5: Balance Q4: Match Pb/CrO₄ atoms
$$\text{Pb(NO}_3)_2 + \text{K}_2(\text{CrO}_4)
ightarrow \text{Pb(CrO}_4) + 2\text{K(NO}_3)$$
Step6: Balance Q5: Match Al atoms
$$2\text{Al(OH)}_3
ightarrow \text{Al}_2\text{O}_3 + \text{H}_2\text{O}$$
Step7: Balance Q5: Match H/O atoms
$$2\text{Al(OH)}_3
ightarrow \text{Al}_2\text{O}_3 + 3\text{H}_2\text{O}$$
Step8: Balance Q6: Match S/Pb atoms
$$\text{PbSO}_3 + \text{O}_2
ightarrow \text{PbSO}_4$$
Step9: Balance Q6: Match O atoms
$$2\text{PbSO}_3 + \text{O}_2
ightarrow 2\text{PbSO}_4$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $3\text{Ca(OH)}_2 + \text{Al}_2(\text{SO}_4)_3
ightarrow 2\text{Al(OH)}_3 + 3\text{Ca(SO}_4)$
- $2\text{NaOH} + \text{H}_2(\text{SO}_4)
ightarrow \text{Na}_2(\text{SO}_4) + 2\text{H}_2\text{O}$
- $\text{Pb(NO}_3)_2 + \text{K}_2(\text{CrO}_4)
ightarrow \text{Pb(CrO}_4) + 2\text{K(NO}_3)$
- $2\text{Al(OH)}_3
ightarrow \text{Al}_2\text{O}_3 + 3\text{H}_2\text{O}$
- $2\text{PbSO}_3 + \text{O}_2
ightarrow 2\text{PbSO}_4$