QUESTION IMAGE
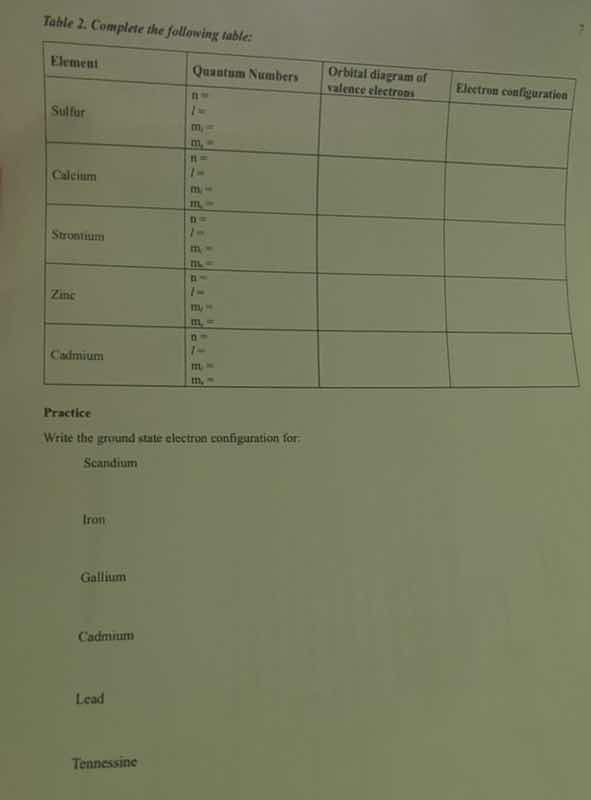
Question
Question was provided via image upload.
Step1: Sulfur (S, Z=16)
Quantum Numbers (valence e⁻: 3s²3p⁴; take a 3p e⁻)
$n=3$ (principal shell), $l=1$ (p orbital), $m_l=+1$ (pₓ orbital), $m_s=+\frac{1}{2}$ (spin up)
Orbital Diagram (valence e⁻):
Electron Configuration:
$1s^22s^22p^63s^23p^4$ or $[Ne]3s^23p^4$
Step2: Calcium (Ca, Z=20)
Quantum Numbers (valence e⁻: 4s²; take a 4s e⁻)
$n=4$, $l=0$ (s orbital), $m_l=0$, $m_s=+\frac{1}{2}$
Orbital Diagram (valence e⁻):
Electron Configuration:
$1s^22s^22p^63s^23p^64s^2$ or $[Ar]4s^2$
Step3: Strontium (Sr, Z=38)
Quantum Numbers (valence e⁻: 5s²; take a 5s e⁻)
$n=5$, $l=0$, $m_l=0$, $m_s=+\frac{1}{2}$
Orbital Diagram (valence e⁻):
Electron Configuration:
$1s^22s^22p^63s^23p^63d^{10}4s^24p^65s^2$ or $[Kr]5s^2$
Step4: Zinc (Zn, Z=30)
Quantum Numbers (valence e⁻: 4s²3d¹⁰; take a 3d e⁻)
$n=3$, $l=2$ (d orbital), $m_l=0$, $m_s=+\frac{1}{2}$
Orbital Diagram (valence e⁻):
Electron Configuration:
$1s^22s^22p^63s^23p^63d^{10}4s^2$ or $[Ar]3d^{10}4s^2$
Step5: Cadmium (Cd, Z=48)
Quantum Numbers (valence e⁻: 5s²4d¹⁰; take a 4d e⁻)
$n=4$, $l=2$, $m_l=0$, $m_s=+\frac{1}{2}$
Orbital Diagram (valence e⁻):
Electron Configuration:
$1s^22s^22p^63s^23p^63d^{10}4s^24p^64d^{10}5s^2$ or $[Kr]4d^{10}5s^2$
Step6: Practice (Ground State Configurations)
Scandium (Sc, Z=21):
Fill orbitals by Aufbau principle: $[Ar]3d^14s^2$
Iron (Fe, Z=26):
Half-fill d orbitals first: $[Ar]3d^64s^2$
Gallium (Ga, Z=31):
Post-transition metal: $[Ar]3d^{10}4s^24p^1$
Cadmium (Cd, Z=48):
As above: $[Kr]4d^{10}5s^2$
Lead (Pb, Z=82):
Post-transition metal: $[Xe]4f^{14}5d^{10}6s^26p^2$
Tennessine (Ts, Z=117):
Halogens, 7th period: $[Rn]5f^{14}6d^{10}7s^27p^5$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Completed Table:
| Element | Quantum Numbers (for one valence e⁻) | Orbital diagram of valence electrons | Electron configuration |
|---|
<br> $3p_x\quad3p_y\quad3p_z$ | $[Ne]3s^23p^4$ |
| Calcium | $n=4, l=0, m_l=0, m_s=+\frac{1}{2}$ |
| $[Ar]4s^2$ |
| Strontium | $n=5, l=0, m_l=0, m_s=+\frac{1}{2}$ |
| $[Kr]5s^2$ |
| Zinc | $n=3, l=2, m_l=0, m_s=+\frac{1}{2}$ |
<br> $3d_{xy}\quad3d_{xz}\quad3d_{yz}\quad3d_{x^2-y^2}\quad3d_{z^2}$ | $[Ar]3d^{10}4s^2$ |
| Cadmium | $n=4, l=2, m_l=0, m_s=+\frac{1}{2}$ |
<br> $4d_{xy}\quad4d_{xz}\quad4d_{yz}\quad4d_{x^2-y^2}\quad4d_{z^2}$ | $[Kr]4d^{10}5s^2$ |
Practice Answers:
- Scandium: $[Ar]3d^14s^2$
- Iron: $[Ar]3d^64s^2$
- Gallium: $[Ar]3d^{10}4s^24p^1$
- Cadmium: $[Kr]4d^{10}5s^2$
- Lead: $[Xe]4f^{14}5d^{10}6s^26p^2$
- Tennessine: $[Rn]5f^{14}6d^{10}7s^27p^5$