QUESTION IMAGE
Question
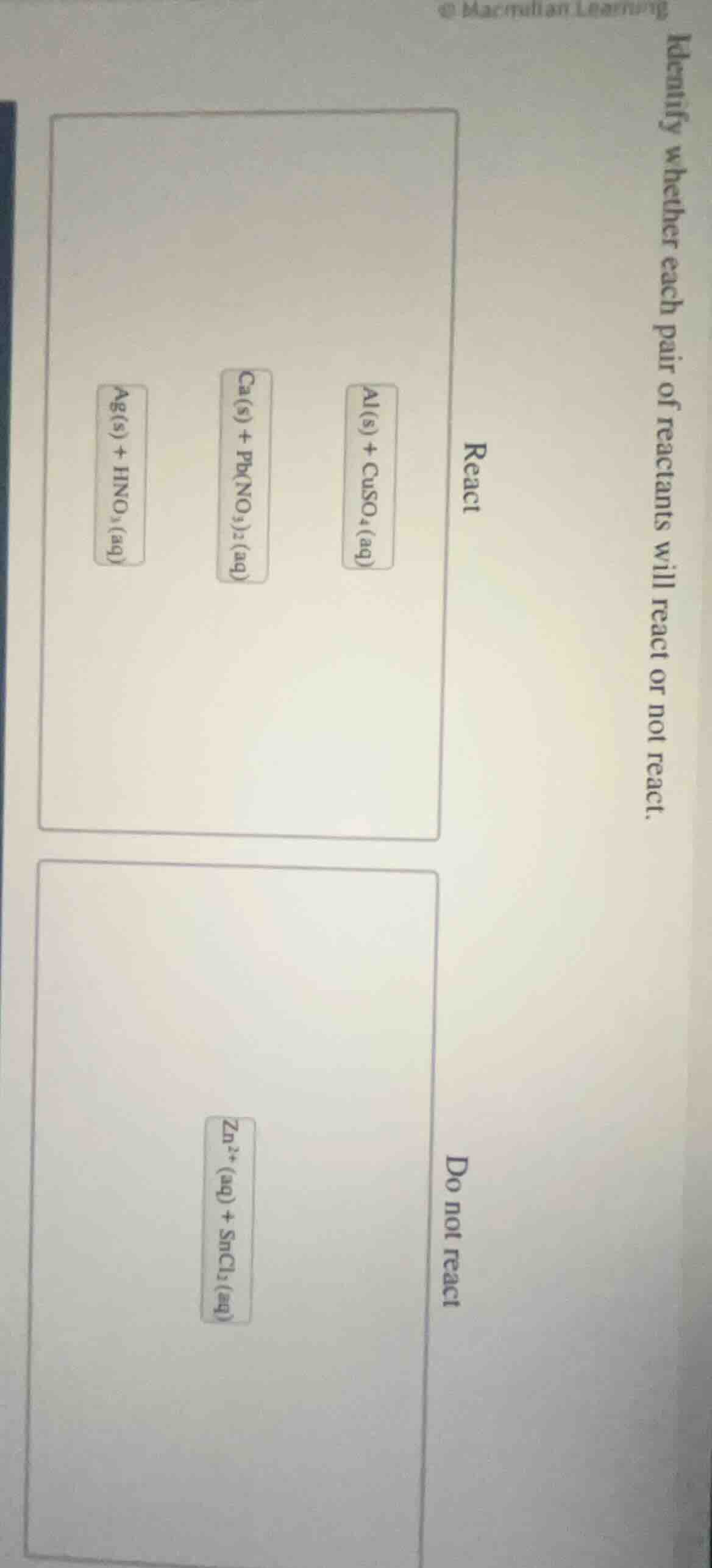
identify whether each pair of reactants will react or not react.
react
al(s) + cuso₄(aq)
ca(s) + pb(no₃)₂(aq)
ag(s) + hno₃(aq)
do not react
zn²⁺(aq) + sncl₂(aq)
Step1: Check metal reactivity for Al+CuSO₄
Al is more reactive than Cu, so single displacement occurs:
Step2: Check metal reactivity for Ca+Pb(NO₃)₂
Ca is more reactive than Pb, so single displacement occurs:
Step3: Check reactivity for Ag+HNO₃
$\text{HNO}_3$ is a strong oxidizing acid, so redox reaction occurs:
Step4: Check reactivity for Zn²⁺+SnCl₂
Zn is more reactive than Sn; $\text{Zn}^{2+}$ cannot oxidize $\text{Sn}^{2+}$, so no reaction.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
React:
$\text{Al}(s) + \text{CuSO}_4(aq)$
$\text{Ca}(s) + \text{Pb(NO}_3\text{)}_2(aq)$
$\text{Ag}(s) + \text{HNO}_3(aq)$
Do not react:
$\text{Zn}^{2+}(aq) + \text{SnCl}_2(aq)$