QUESTION IMAGE
Question
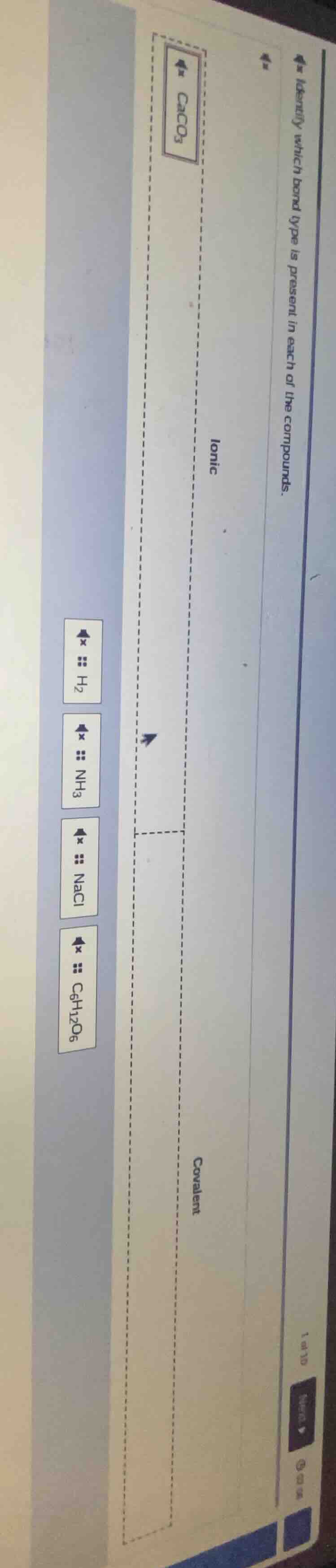
identify which bond type is present in each of the compounds.
caco₃
ionic
covalent
h₂
nh₃
nacl
c₆h₁₂o₆
Response
To determine the bond type (ionic or covalent) for each compound, we analyze the nature of the atoms involved:
1. \( \boldsymbol{\ce{CaCO3}} \)
- Ionic Bond: Calcium (\( \ce{Ca} \)) is a metal, and the carbonate ion (\( \ce{CO3^{2-}} \)) is a polyatomic ion. Metal-nonmetal (or metal-polyatomic ion) interactions form ionic bonds.
- Covalent Bond (within \( \boldsymbol{\ce{CO3^{2-}}} \)): Carbon and oxygen are nonmetals, so the bonds within the carbonate ion are covalent. However, \( \ce{CaCO3} \) is classified as an ionic compound overall (due to the bond between \( \ce{Ca^{2+}} \) and \( \ce{CO3^{2-}} \)).
2. \( \boldsymbol{\ce{H2}} \)
- Covalent Bond: Hydrogen is a nonmetal, and \( \ce{H2} \) consists of two identical nonmetal atoms sharing electrons (nonpolar covalent bond).
3. \( \boldsymbol{\ce{NH3}} \)
- Covalent Bond: Nitrogen and hydrogen are both nonmetals. They share electrons to form covalent bonds (polar covalent, due to electronegativity differences).
4. \( \boldsymbol{\ce{NaCl}} \)
- Ionic Bond: Sodium (\( \ce{Na} \), a metal) transfers an electron to chlorine (\( \ce{Cl} \), a nonmetal), forming \( \ce{Na^+} \) and \( \ce{Cl^-} \) ions held by ionic attraction.
5. \( \boldsymbol{\ce{C6H12O6}} \) (Glucose)
- Covalent Bond: Carbon, hydrogen, and oxygen are all nonmetals. The bonds between these atoms (e.g., \( \ce{C-C} \), \( \ce{C-H} \), \( \ce{C-O} \)) are covalent (electron sharing).
Final Classification:
| Compound | Bond Type | Reason |
|---|---|---|
| \( \ce{H2} \) | Covalent | Two nonmetal atoms (H) sharing electrons. |
| \( \ce{NH3} \) | Covalent | Nonmetals (N, H) sharing electrons. |
| \( \ce{NaCl} \) | Ionic | Metal (Na) + nonmetal (Cl) (electron transfer). |
| \( \ce{C6H12O6} \) | Covalent | Nonmetals (C, H, O) sharing electrons. |
If you need to drag and drop:
- Ionic Column: \( \ce{CaCO3} \), \( \ce{NaCl} \)
- Covalent Column: \( \ce{H2} \), \( \ce{NH3} \), \( \ce{C6H12O6} \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To determine the bond type (ionic or covalent) for each compound, we analyze the nature of the atoms involved:
1. \( \boldsymbol{\ce{CaCO3}} \)
- Ionic Bond: Calcium (\( \ce{Ca} \)) is a metal, and the carbonate ion (\( \ce{CO3^{2-}} \)) is a polyatomic ion. Metal-nonmetal (or metal-polyatomic ion) interactions form ionic bonds.
- Covalent Bond (within \( \boldsymbol{\ce{CO3^{2-}}} \)): Carbon and oxygen are nonmetals, so the bonds within the carbonate ion are covalent. However, \( \ce{CaCO3} \) is classified as an ionic compound overall (due to the bond between \( \ce{Ca^{2+}} \) and \( \ce{CO3^{2-}} \)).
2. \( \boldsymbol{\ce{H2}} \)
- Covalent Bond: Hydrogen is a nonmetal, and \( \ce{H2} \) consists of two identical nonmetal atoms sharing electrons (nonpolar covalent bond).
3. \( \boldsymbol{\ce{NH3}} \)
- Covalent Bond: Nitrogen and hydrogen are both nonmetals. They share electrons to form covalent bonds (polar covalent, due to electronegativity differences).
4. \( \boldsymbol{\ce{NaCl}} \)
- Ionic Bond: Sodium (\( \ce{Na} \), a metal) transfers an electron to chlorine (\( \ce{Cl} \), a nonmetal), forming \( \ce{Na^+} \) and \( \ce{Cl^-} \) ions held by ionic attraction.
5. \( \boldsymbol{\ce{C6H12O6}} \) (Glucose)
- Covalent Bond: Carbon, hydrogen, and oxygen are all nonmetals. The bonds between these atoms (e.g., \( \ce{C-C} \), \( \ce{C-H} \), \( \ce{C-O} \)) are covalent (electron sharing).
Final Classification:
| Compound | Bond Type | Reason |
|---|---|---|
| \( \ce{H2} \) | Covalent | Two nonmetal atoms (H) sharing electrons. |
| \( \ce{NH3} \) | Covalent | Nonmetals (N, H) sharing electrons. |
| \( \ce{NaCl} \) | Ionic | Metal (Na) + nonmetal (Cl) (electron transfer). |
| \( \ce{C6H12O6} \) | Covalent | Nonmetals (C, H, O) sharing electrons. |
If you need to drag and drop:
- Ionic Column: \( \ce{CaCO3} \), \( \ce{NaCl} \)
- Covalent Column: \( \ce{H2} \), \( \ce{NH3} \), \( \ce{C6H12O6} \)