QUESTION IMAGE
Question
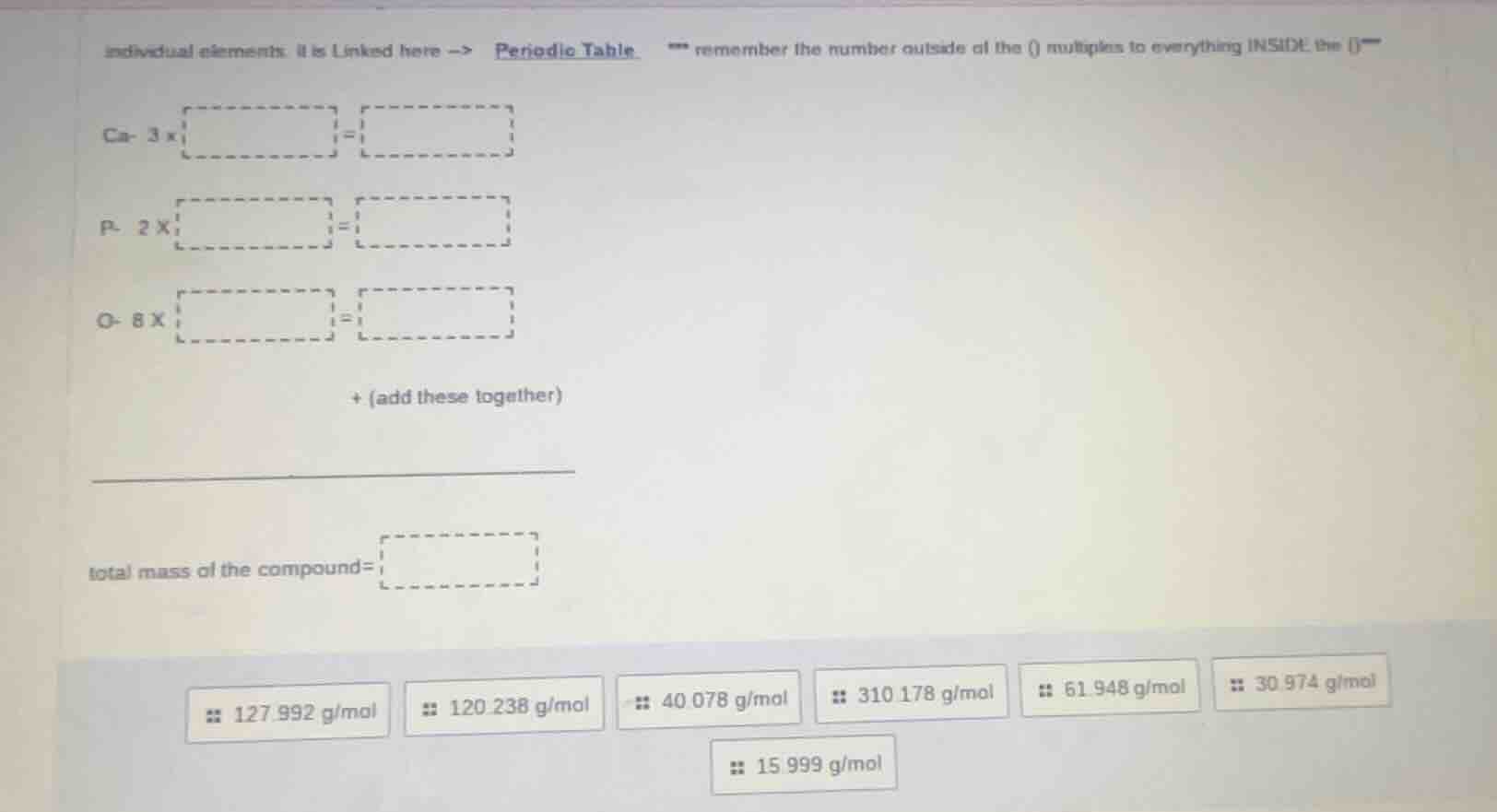
individual elements. it is linked here -> periodic table. remember the number outside of the () multiplies to everything inside the ()
ca- 3 x =
p- 2 x =
o- 8 x =
+ (add these together)
_________________________
total mass of the compound=
127.992 g/mol 120.238 g/mol 40.078 g/mol 310.178 g/mol 61.948 g/mol 30.974 g/mol
15.999 g/mol
Step1: Calculate mass for Ca
The molar mass of Ca is $40.078$ g/mol. We have 3 moles of Ca, so the mass is $3\times40.078$.
$3\times40.078 = 120.234$ (close to 120.238 g/mol, considering possible rounding)
Step2: Calculate mass for P
The molar mass of P is $30.974$ g/mol. We have 2 moles of P, so the mass is $2\times30.974$.
$2\times30.974 = 61.948$ g/mol
Step3: Calculate mass for O
The molar mass of O is $15.999$ g/mol. We have 8 moles of O, so the mass is $8\times15.999$.
$8\times15.999 = 127.992$ g/mol
Step4: Sum the masses
Now we add the masses of Ca, P, and O: $120.238 + 61.948 + 127.992$.
$120.238+61.948 = 182.186$; $182.186 + 127.992 = 310.178$ g/mol
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The total mass of the compound is $310.178$ g/mol.
For each element:
- Ca: $3\times40.078 = 120.238$ g/mol
- P: $2\times30.974 = 61.948$ g/mol
- O: $8\times15.999 = 127.992$ g/mol