QUESTION IMAGE
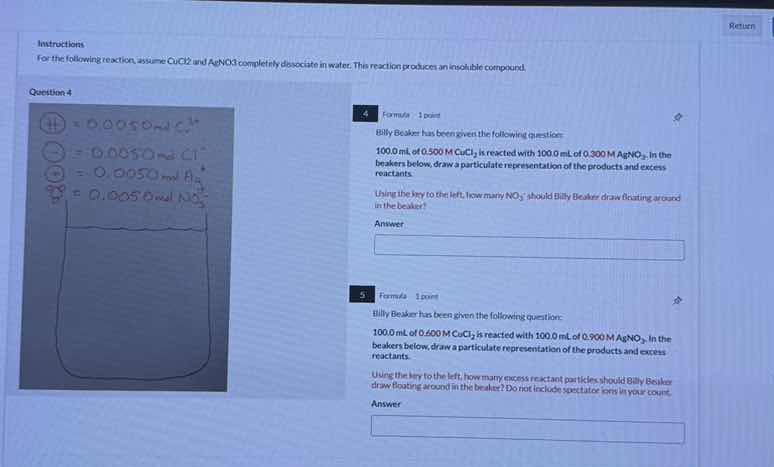
Question
instructions
for the following reaction, assume cucl₂ and agno₃ completely dissociate in water. this reaction produces an insoluble compound.
question 4
4 formula 1 point
billy beaker has been given the following question:
100.0 ml of 0.0500 m cucl₂ is reacted with 100.0 ml of 0.0300 m agno₃. in the beaker below, draw a particulate representation of the products and excess reactants.
using the key to the left, how many no₃⁻ should billy beaker draw floating around in the beaker?
answer
5 formula 1 point
billy beaker has been given the following question:
100.0 ml of 0.0500 m cucl₂ is reacted with 100.0 ml of 0.0300 m agno₃. in the beaker below, draw a particulate representation of the products and excess reactants.
using the key to the left, how many more excess reactant particles should billy beaker draw floating around in the beaker? do not include spectator ions in your count.
answer
Step1: Calculate moles of $\text{Cl}^-$
Moles of $\text{CuCl}_2 = 0.300\ \text{mol/L} \times 0.100\ \text{L} = 0.0300\ \text{mol}$
Each $\text{CuCl}_2$ gives 2 $\text{Cl}^-$, so moles of $\text{Cl}^- = 2 \times 0.0300\ \text{mol} = 0.0600\ \text{mol}$
Step2: Calculate moles of $\text{Ag}^+$
Moles of $\text{AgNO}_3 = 0.300\ \text{mol/L} \times 0.100\ \text{L} = 0.0300\ \text{mol}$
Each $\text{AgNO}_3$ gives 1 $\text{Ag}^+$, so moles of $\text{Ag}^+ = 0.0300\ \text{mol}$
Step3: Find moles of $\text{NO}_3^-$
$\text{NO}_3^-$ is a spectator ion, so moles of $\text{NO}_3^-$ = moles of $\text{AgNO}_3 = 0.0300\ \text{mol}$
From the key, 0.0050 mol = 1 symbol. Number of $\text{NO}_3^-$ symbols = $\frac{0.0300\ \text{mol}}{0.0050\ \text{mol/symbol}} = 6$
---
Step4: Calculate moles of $\text{Cl}^-$ (Q5)
Moles of $\text{CuCl}_2 = 0.600\ \text{mol/L} \times 0.100\ \text{L} = 0.0600\ \text{mol}$
Moles of $\text{Cl}^- = 2 \times 0.0600\ \text{mol} = 0.120\ \text{mol}$
Step5: Calculate moles of $\text{Ag}^+$ (Q5)
Moles of $\text{AgNO}_3 = 0.600\ \text{mol/L} \times 0.100\ \text{L} = 0.0600\ \text{mol}$
Moles of $\text{Ag}^+ = 0.0600\ \text{mol}$
Step6: Find excess $\text{Cl}^-$ (Q5)
Reaction: $\text{Ag}^+ + \text{Cl}^-
ightarrow \text{AgCl}(s)$. 1:1 ratio, so excess $\text{Cl}^- = 0.120\ \text{mol} - 0.0600\ \text{mol} = 0.0600\ \text{mol}$
Number of excess $\text{Cl}^-$ symbols = $\frac{0.0600\ \text{mol}}{0.0050\ \text{mol/symbol}} = 12$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 4: 6
Question 5: 12