QUESTION IMAGE
Question
intermolecular forces
quiz complete
1 2 3 4 5 6 7 8 9 10
time remain
58:10
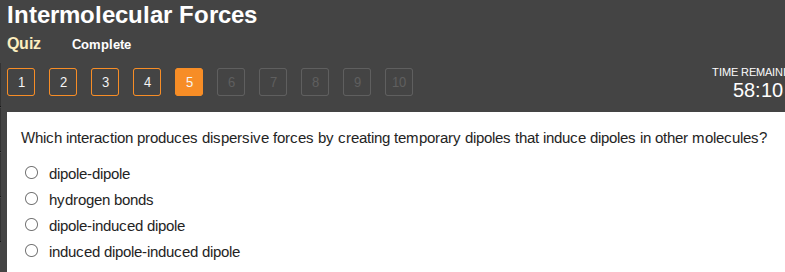
which interaction produces dispersive forces by creating temporary dipoles that induce dipoles in other molecules?
dipole-dipole
hydrogen bonds
dipole-induced dipole
induced dipole-induced dipole
Dispersive (London dispersion) forces arise from temporary dipoles (induced dipoles) in molecules, which then induce dipoles in adjacent molecules. This matches the description of induced dipole-induced dipole interactions. Dipole-dipole forces involve permanent dipoles, hydrogen bonds are a strong dipole-dipole subset, and dipole-induced dipole requires one permanent dipole and one induced dipole.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
induced dipole-induced dipole