QUESTION IMAGE
Question
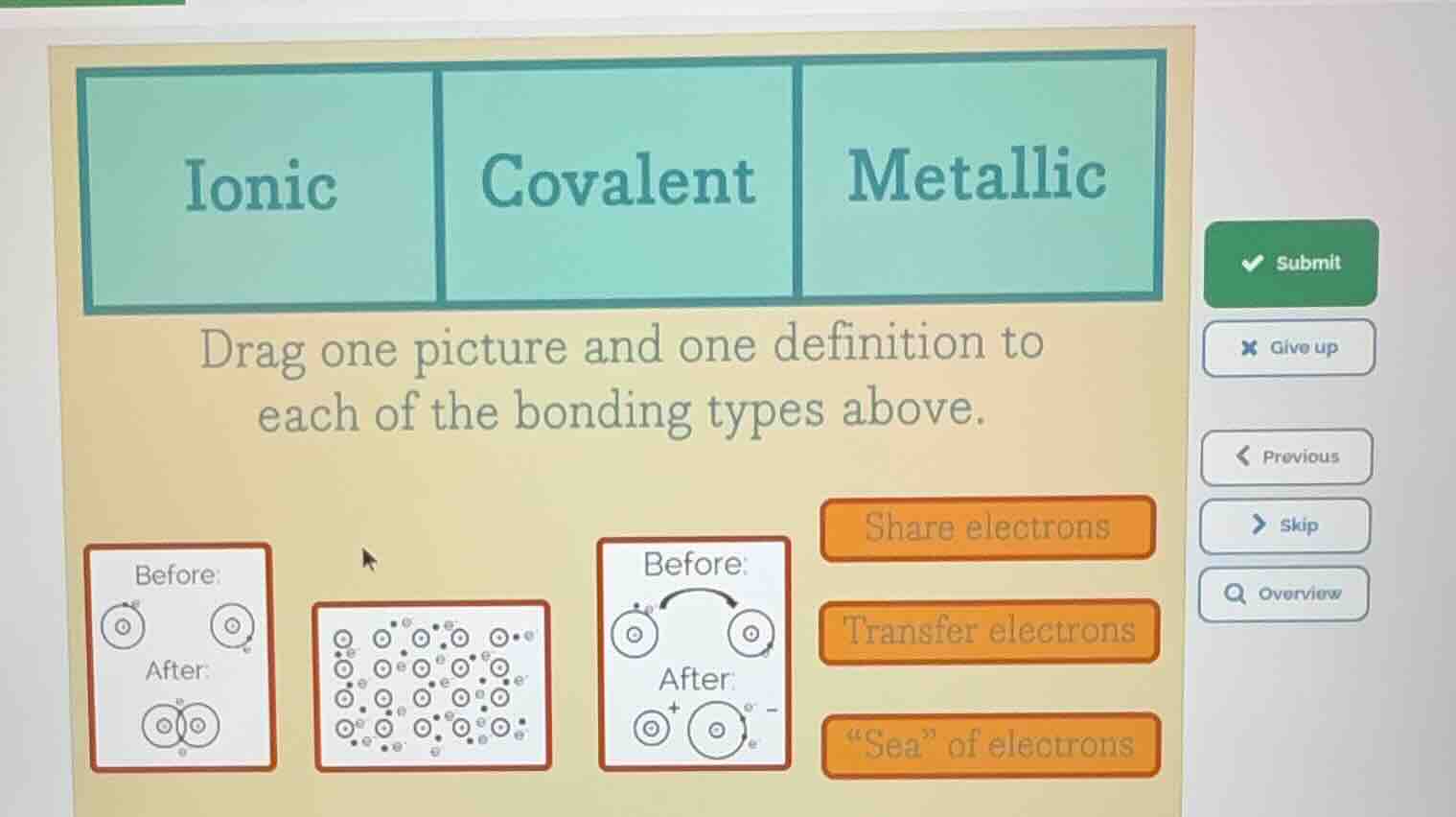
ionic covalent metallic drag one picture and one definition to each of the bonding types above. before: after: before: after: before: after: share electrons transfer electrons \sea\ of electrons
Brief Explanations
- Ionic Bonding: Involves the transfer of electrons from one atom to another (usually between a metal and a non - metal) to form ions. The diagram with electron transfer (the one where electrons move from one atom to another, resulting in charged ions) matches this, and the definition is "Transfer electrons".
- Covalent Bonding: Atoms share electrons to achieve a stable electron configuration (usually between non - metals). The diagram where atoms share electrons (the one with the overlapping orbitals or shared electron pair) matches, and the definition is "Share electrons".
- Metallic Bonding: Consists of a "sea" of delocalized electrons surrounding positively charged metal ions. The diagram with a lattice of metal atoms and a "sea" of electrons (the one with many atoms and free - moving electrons) matches, and the definition is "‘Sea’ of electrons".
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Ionic: Drag the diagram with electron transfer (the one labeled with Before and After showing electron transfer to form ions) and the "Transfer electrons" definition.
- Covalent: Drag the diagram with shared electrons (the one with overlapping or shared electron representation) and the "Share electrons" definition.
- Metallic: Drag the diagram with the lattice of metal atoms and free electrons (the one with many atoms and a "sea" - like electron representation) and the "‘Sea’ of electrons" definition.