QUESTION IMAGE
Question
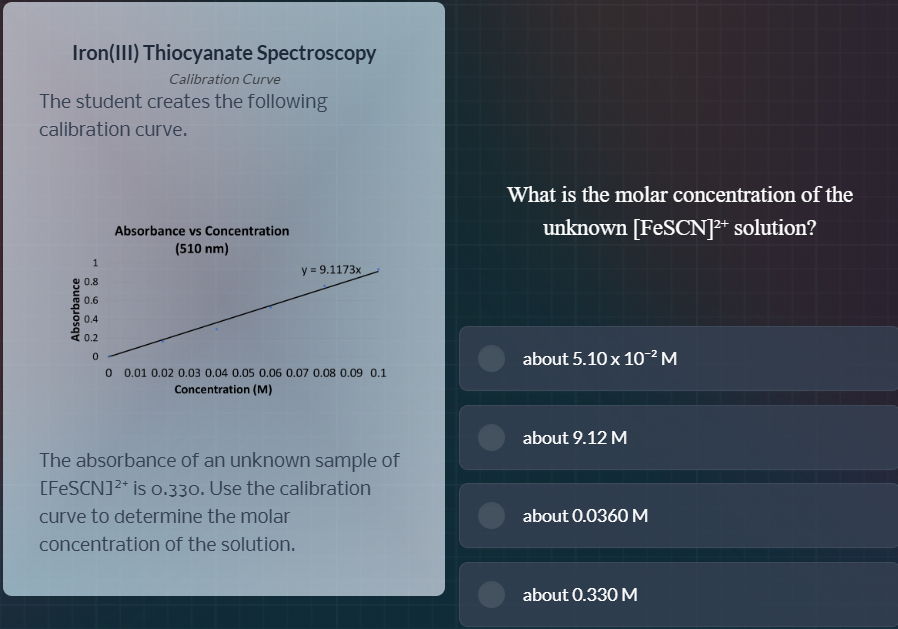
iron(iii) thiocyanate spectroscopycalibration curvethe student creates the following calibration curve.absorbance vs concentration(510 nm)y = 9.1173xthe absorbance of an unknown sample of fescn²⁺ is 0.330. use the calibration curve to determine the molar concentration of the solution.what is the molar concentration of the unknown fescn²⁺ solution?about 5.10 x 10⁻² mabout 9.12 mabout 0.0360 mabout 0.330 m
Step1: Define given variables
Given $y=0.330$ (absorbance), $y=9.1173x$ (calibration equation)
Step2: Solve for $x$ (concentration)
Rearrange equation: $x=\frac{y}{9.1173}$
Substitute values: $x=\frac{0.330}{9.1173}$
Step3: Calculate concentration
$x\approx0.0362$ M, rounded to 0.0360 M
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
about 0.0360 M