QUESTION IMAGE
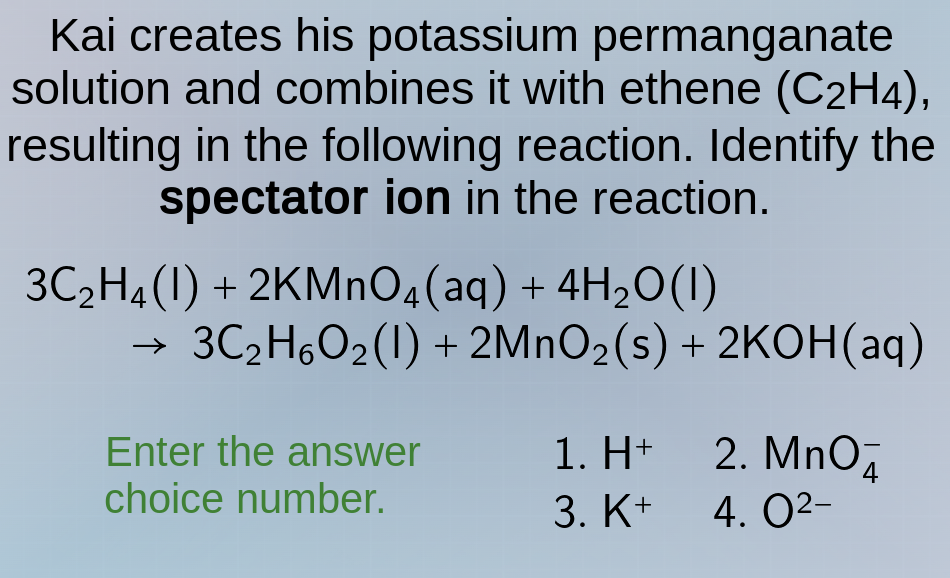
Question
kai creates his potassium permanganate solution and combines it with ethene (c₂h₄), resulting in the following reaction. identify the spectator ion in the reaction.
3c₂h₄(l) + 2kmno₄(aq) + 4h₂o(l) → 3c₂h₆o₂(l) + 2mno₂(s) + 2koh(aq)
enter the answer choice number.
- h⁺ 2. mno₄⁻
- k⁺ 4. o²⁻
Brief Explanations
A spectator ion is an ion that does not participate in the chemical reaction and remains in the same form on both sides of the reaction. Let's analyze the reaction:
- For \( \ce{KMnO4(aq)} \), it dissociates into \( \ce{K+} \) and \( \ce{MnO4-} \) in aqueous solution.
- For \( \ce{KOH(aq)} \), it dissociates into \( \ce{K+} \) and \( \ce{OH-} \) in aqueous solution.
Looking at the ions:
- \( \ce{MnO4-} \) is involved in the reaction (it is reduced to \( \ce{MnO2} \)), so it is not a spectator ion.
- \( \ce{H+} \) is not present as a free ion in the given reactants or products in the way to be a spectator (the reaction involves water and formation of \( \ce{KOH} \), not free \( \ce{H+} \) in the relevant sense here).
- \( \ce{O^{2-}} \) is not a free ion in this aqueous reaction context (oxides like \( \ce{O^{2-}} \) are not present as free ions in these aqueous solutions).
- \( \ce{K+} \) is present in \( \ce{KMnO4} \) (as \( \ce{K+} \)) and in \( \ce{KOH} \) (as \( \ce{K+} \)), and it does not undergo any chemical change (it is just a cation that stays as \( \ce{K+} \) throughout the reaction, not participating in the redox or other chemical changes). So \( \ce{K+} \) is the spectator ion.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- \( \ce{K+} \)