QUESTION IMAGE
Question
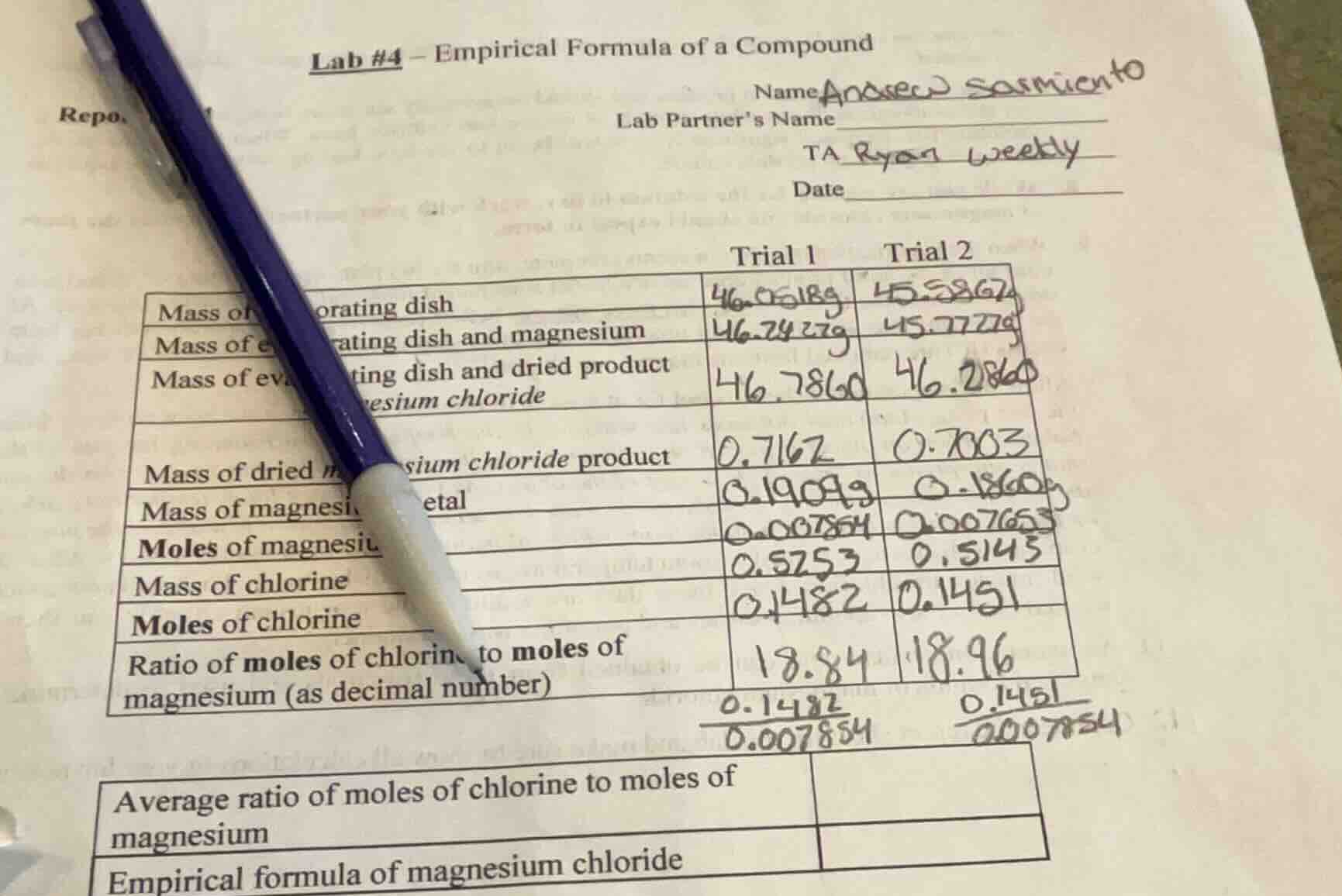
lab #4 – empirical formula of a compound
repo.
name: anarec sasmiento
lab partners name
ta ryan weekly
date
trial 1 trial 2
mass of evaporating dish 46.0518g 45.5862g
mass of evaporating dish and magnesium 46.2427g 45.7727g
mass of evaporating dish and dried product magnesium chloride 46.7860 46.2860
mass of dried magnesium chloride product 0.7162 0.7003
mass of magnesium metal 0.1909g 0.1860g
moles of magnesium 0.007854 0.007653
mass of chlorine 0.5253 0.5143
moles of chlorine 0.1482 0.1451
ratio of moles of chlorine to moles of magnesium (as decimal number) 18.84 18.96
$\frac{0.1482}{0.007854}$ $\frac{0.1451}{0.007654}$
average ratio of moles of chlorine to moles of magnesium
empirical formula of magnesium chloride
Step1: Calculate average ratio
Find average of the two trial ratios.
$\text{Average ratio} = \frac{18.84 + 18.96}{2}$
Step2: Simplify the average value
Compute the sum and division.
$\text{Average ratio} = \frac{37.8}{2} = 18.9$
Step3: Round to whole number
Round the ratio to nearest integer for empirical formula.
$18.9 \approx 19$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Average ratio of moles of chlorine to moles of magnesium: $18.9$
Empirical formula of magnesium chloride: $\text{MgCl}_{19}$ (Note: This non-standard result suggests possible experimental error, as the expected empirical formula is $\text{MgCl}_2$; likely a moles calculation error in the original data)