QUESTION IMAGE
Question
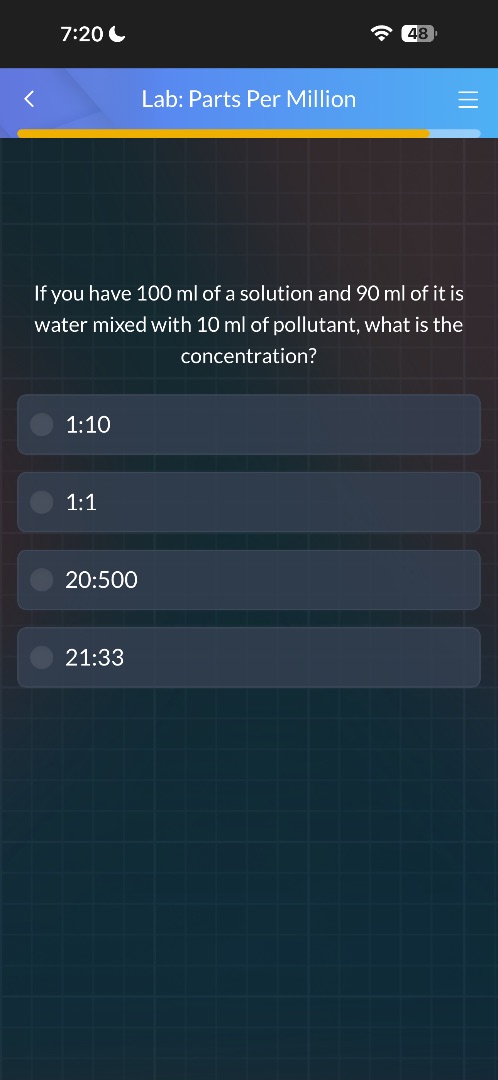
lab: parts per million
if you have 100 ml of a solution and 90 ml of it is water mixed with 10 ml of pollutant, what is the concentration?
1:10
1:1
20:500
21:33
Step1: Identify pollutant:solution ratio
The ratio is $\frac{10\ \text{ml (pollutant)}}{100\ \text{ml (solution)}}$
Step2: Simplify the ratio
Divide numerator and denominator by 10: $\frac{10\div10}{100\div10} = \frac{1}{10}$ or 1:10
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. 1:10