QUESTION IMAGE
Question
labels ① drop zones ← reset all image description
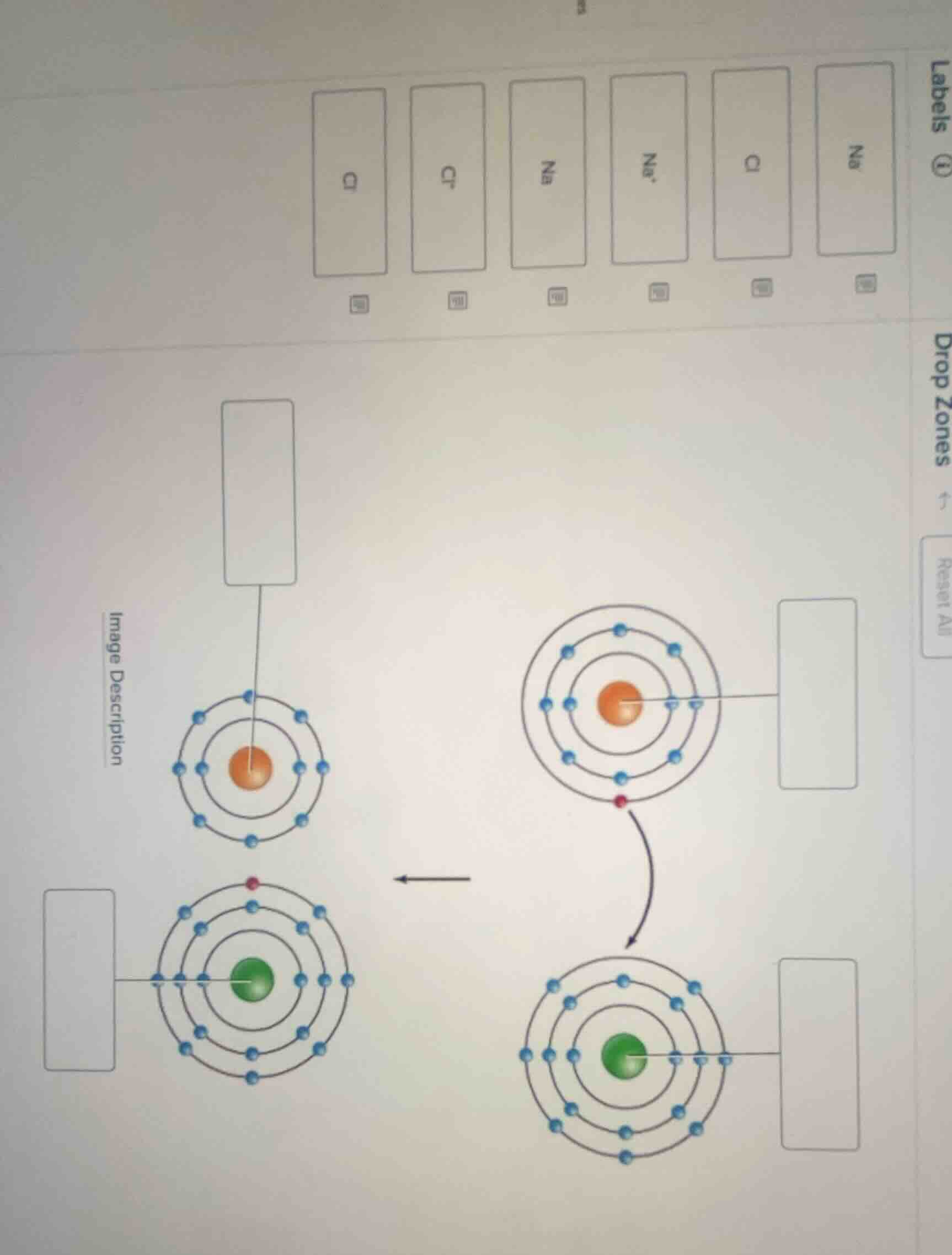
To solve this problem, we analyze the electron - proton configurations of sodium (Na) and chlorine (Cl) atoms and their ions:
Step 1: Analyze the sodium atom/ion
- A sodium (Na) atom has 11 protons and 11 electrons. Its electron configuration is \(2, 8, 1\). When it forms a sodium ion (\(Na^+\)), it loses one electron, so the electron configuration becomes \(2, 8\), and the number of protons remains 11.
- Looking at the orange - centered (representing sodium - related) structures:
- The structure with the electron configuration \(2, 8, 1\) (has three electron shells, with 1 electron in the outermost shell) should be labeled \(Na\) (sodium atom).
- The structure with the electron configuration \(2, 8\) (has two electron shells, since it lost one electron) should be labeled \(Na^+\) (sodium ion). And the center (nucleus) of the sodium - related structures, which has 11 protons, is still related to sodium, so the nucleus of the sodium atom (with 11 protons) is part of the \(Na\) atom representation, and the nucleus of the sodium ion (also with 11 protons) is part of the \(Na^+\) ion representation.
Step 2: Analyze the chlorine atom/ion
- A chlorine (Cl) atom has 17 protons and 17 electrons. Its electron configuration is \(2, 8, 7\). When it forms a chloride ion (\(Cl^-\)), it gains one electron, so the electron configuration becomes \(2, 8, 8\), and the number of protons remains 17.
- Looking at the green - centered (representing chlorine - related) structures:
- The structure with the electron configuration \(2, 8, 7\) (has three electron shells, with 7 electrons in the outermost shell) should be labeled \(Cl\) (chlorine atom).
- The structure with the electron configuration \(2, 8, 8\) (has three electron shells, since it gained one electron) should be labeled \(Cl^-\) (chloride ion). And the center (nucleus) of the chlorine - related structures, which has 17 protons, is part of the \(Cl\) atom or \(Cl^-\) ion representation.
Labeling the drop - zones:
- For the orange - centered structure with electron configuration \(2, 8, 1\) (three electron shells, 1 electron in the outermost shell): Label it \(Na\).
- For the orange - centered structure with electron configuration \(2, 8\) (two electron shells): Label it \(Na^+\).
- For the green - centered structure with electron configuration \(2, 8, 7\) (three electron shells, 7 electrons in the outermost shell): Label it \(Cl\).
- For the green - centered structure with electron configuration \(2, 8, 8\) (three electron shells, 8 electrons in the outermost shell): Label it \(Cl^-\).
- For the nucleus of the sodium - related (orange - centered) structures (which has 11 protons, characteristic of sodium): It is part of the \(Na\) (when it's the atom) or \(Na^+\) (when it's the ion) representation, so the nucleus label is related to sodium, and the atom/ion labels are as above.
- For the nucleus of the chlorine - related (green - centered) structures (which has 17 protons, characteristic of chlorine): It is part of the \(Cl\) (when it's the atom) or \(Cl^-\) (when it's the ion) representation, so the nucleus label is related to chlorine, and the atom/ion labels are as above.
Final Labeling (assuming the drop - zones are ordered from top - left, top - right, bottom - left, bottom - right for the atom/ion structures and their nuclei):
- Top - left (orange - centered, \(2, 8, 1\)): \(Na\)
- Top - right (nucleus of orange - centered): \(Na\) (nucleus of sodium atom)
- Bottom - left (green - centered, \(2, 8, 7\)): \(Cl\)
- Bottom - right (green - centered, \(2,…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this problem, we analyze the electron - proton configurations of sodium (Na) and chlorine (Cl) atoms and their ions:
Step 1: Analyze the sodium atom/ion
- A sodium (Na) atom has 11 protons and 11 electrons. Its electron configuration is \(2, 8, 1\). When it forms a sodium ion (\(Na^+\)), it loses one electron, so the electron configuration becomes \(2, 8\), and the number of protons remains 11.
- Looking at the orange - centered (representing sodium - related) structures:
- The structure with the electron configuration \(2, 8, 1\) (has three electron shells, with 1 electron in the outermost shell) should be labeled \(Na\) (sodium atom).
- The structure with the electron configuration \(2, 8\) (has two electron shells, since it lost one electron) should be labeled \(Na^+\) (sodium ion). And the center (nucleus) of the sodium - related structures, which has 11 protons, is still related to sodium, so the nucleus of the sodium atom (with 11 protons) is part of the \(Na\) atom representation, and the nucleus of the sodium ion (also with 11 protons) is part of the \(Na^+\) ion representation.
Step 2: Analyze the chlorine atom/ion
- A chlorine (Cl) atom has 17 protons and 17 electrons. Its electron configuration is \(2, 8, 7\). When it forms a chloride ion (\(Cl^-\)), it gains one electron, so the electron configuration becomes \(2, 8, 8\), and the number of protons remains 17.
- Looking at the green - centered (representing chlorine - related) structures:
- The structure with the electron configuration \(2, 8, 7\) (has three electron shells, with 7 electrons in the outermost shell) should be labeled \(Cl\) (chlorine atom).
- The structure with the electron configuration \(2, 8, 8\) (has three electron shells, since it gained one electron) should be labeled \(Cl^-\) (chloride ion). And the center (nucleus) of the chlorine - related structures, which has 17 protons, is part of the \(Cl\) atom or \(Cl^-\) ion representation.
Labeling the drop - zones:
- For the orange - centered structure with electron configuration \(2, 8, 1\) (three electron shells, 1 electron in the outermost shell): Label it \(Na\).
- For the orange - centered structure with electron configuration \(2, 8\) (two electron shells): Label it \(Na^+\).
- For the green - centered structure with electron configuration \(2, 8, 7\) (three electron shells, 7 electrons in the outermost shell): Label it \(Cl\).
- For the green - centered structure with electron configuration \(2, 8, 8\) (three electron shells, 8 electrons in the outermost shell): Label it \(Cl^-\).
- For the nucleus of the sodium - related (orange - centered) structures (which has 11 protons, characteristic of sodium): It is part of the \(Na\) (when it's the atom) or \(Na^+\) (when it's the ion) representation, so the nucleus label is related to sodium, and the atom/ion labels are as above.
- For the nucleus of the chlorine - related (green - centered) structures (which has 17 protons, characteristic of chlorine): It is part of the \(Cl\) (when it's the atom) or \(Cl^-\) (when it's the ion) representation, so the nucleus label is related to chlorine, and the atom/ion labels are as above.
Final Labeling (assuming the drop - zones are ordered from top - left, top - right, bottom - left, bottom - right for the atom/ion structures and their nuclei):
- Top - left (orange - centered, \(2, 8, 1\)): \(Na\)
- Top - right (nucleus of orange - centered): \(Na\) (nucleus of sodium atom)
- Bottom - left (green - centered, \(2, 8, 7\)): \(Cl\)
- Bottom - right (green - centered, \(2, 8, 8\)): \(Cl^-\)
- And for the other labels:
- The structure with the electron configuration \(2, 8\) (orange - centered, two electron shells): \(Na^+\)
- The nucleus of the green - centered (chlorine - related) structures: For the \(Cl\) atom nucleus, it's part of \(Cl\), and for the \(Cl^-\) ion nucleus, it's part of \(Cl^-\)
If we consider the labels given (\(Cl\), \(Cl^-\), \(Na\), \(Na^+\), \(Cl\), \(Na\)) and match them to the structures:
- The sodium atom (with \(2, 8, 1\)): \(Na\)
- The sodium ion (with \(2, 8\)): \(Na^+\)
- The chlorine atom (with \(2, 8, 7\)): \(Cl\)
- The chloride ion (with \(2, 8, 8\)): \(Cl^-\)
- The nucleus of sodium (in both atom and ion): related to \(Na\) or \(Na^+\) (since nucleus has 11 protons)
- The nucleus of chlorine (in both atom and ion): related to \(Cl\) or \(Cl^-\) (since nucleus has 17 protons)
So, for example, if we have to label the structures:
- The orange - centered structure with 3 electron shells (1 electron in outer shell): \(Na\)
- The orange - centered structure with 2 electron shells: \(Na^+\)
- The green - centered structure with 3 electron shells (7 electrons in outer shell): \(Cl\)
- The green - centered structure with 3 electron shells (8 electrons in outer shell): \(Cl^-\)
- The nucleus of the orange - centered (sodium) structures: \(Na\) (nucleus)
- The nucleus of the green - centered (chlorine) structures: \(Cl\) (nucleus)
The final labeling (matching the given labels to the drop - zones) would be:
- For the structure with the electron configuration of sodium atom (\(2, 8, 1\)): \(Na\)
- For the structure with the electron configuration of sodium ion (\(2, 8\)): \(Na^+\)
- For the structure with the electron configuration of chlorine atom (\(2, 8, 7\)): \(Cl\)
- For the structure with the electron configuration of chloride ion (\(2, 8, 8\)): \(Cl^-\)
- And the nuclei:
- Sodium nucleus: \(Na\)
- Chlorine nucleus: \(Cl\)
If we list the answers for each drop - zone (assuming 4 main drop - zones for the atom/ion structures and 2 for the nuclei, but based on the image):
- Drop - zone 1 (orange - centered, \(2, 8, 1\)): \(Na\)
- Drop - zone 2 (nucleus of orange - centered): \(Na\)
- Drop - zone 3 (green - centered, \(2, 8, 7\)): \(Cl\)
- Drop - zone 4 (green - centered, \(2, 8, 8\)): \(Cl^-\)
- And for the other two drop - zones (the remaining labels):
- One is \(Na^+\) (sodium ion)
- One is \(Cl\) (chlorine atom or its nucleus) or \(Cl^-\) (chloride ion)
A more precise answer, considering the formation of sodium chloride (ionic bond, where \(Na\) loses an electron to \(Cl\) to form \(Na^+\) and \(Cl^-\)):
- The sodium atom (before losing electron): \(Na\)
- The sodium ion (after losing electron): \(Na^+\)
- The chlorine atom (before gaining electron): \(Cl\)
- The chloride ion (after gaining electron): \(Cl^-\)
- The nucleus of sodium (in both atom and ion): \(Na\) (since number of protons doesn't change)
- The nucleus of chlorine (in both atom and ion): \(Cl\) (since number of protons doesn't change)
So if we have to assign the labels:
- Structure with \(Na\) atom (electron config \(2,8,1\)): \(Na\)
- Structure with \(Na^+\) ion (electron config \(2,8\)): \(Na^+\)
- Structure with \(Cl\) atom (electron config \(2,8,7\)): \(Cl\)
- Structure with \(Cl^-\) ion (electron config \(2,8,8\)): \(Cl^-\)
- Nucleus of \(Na\) (atom or ion): \(Na\)
- Nucleus of \(Cl\) (atom or ion): \(Cl\)
The answer depends on the exact drop - zone mapping, but the key is to identify the electron configurations and match them to the atom/ion labels.
If we assume the drop - zones are for the four main structures (two sodium - related, two chlorine - related) and their two nuclei:
- Sodium atom: \(Na\)
- Sodium ion: \(Na^+\)
- Chlorine atom: \(Cl\)
- Chloride ion: \(Cl^-\)
- Sodium nucleus: \(Na\)
- Chlorine nucleus: \(Cl\)
So, for example, if we have to put the labels in the drop - zones:
- \(Na\) (sodium atom)
- \(Na\) (sodium nucleus)
- \(Cl\) (chlorine atom)
- \(Cl^-\) (chloride ion)
- \(Na^+\) (sodium ion)
- \(Cl\) (chlorine nucleus)
(The exact assignment may vary slightly depending on the visual details of the image, but the above is the general principle based on electron configurations and ion formation.)
Final Answer (assuming the drop - zones are labeled from left to right, top to bottom for the structures and their nuclei):
- Top - left: \(Na\)
- Top - middle: \(Na^+\)
- Top - right: \(Na\) (nucleus)
- Bottom - left: \(Cl\)
- Bottom - middle: \(Cl^-\)
- Bottom - right: \(Cl\) (nucleus)
(Or a similar assignment based on the actual image's drop - zone positions. The key is to match the electron configurations to the atom/ion labels.)