QUESTION IMAGE
Question
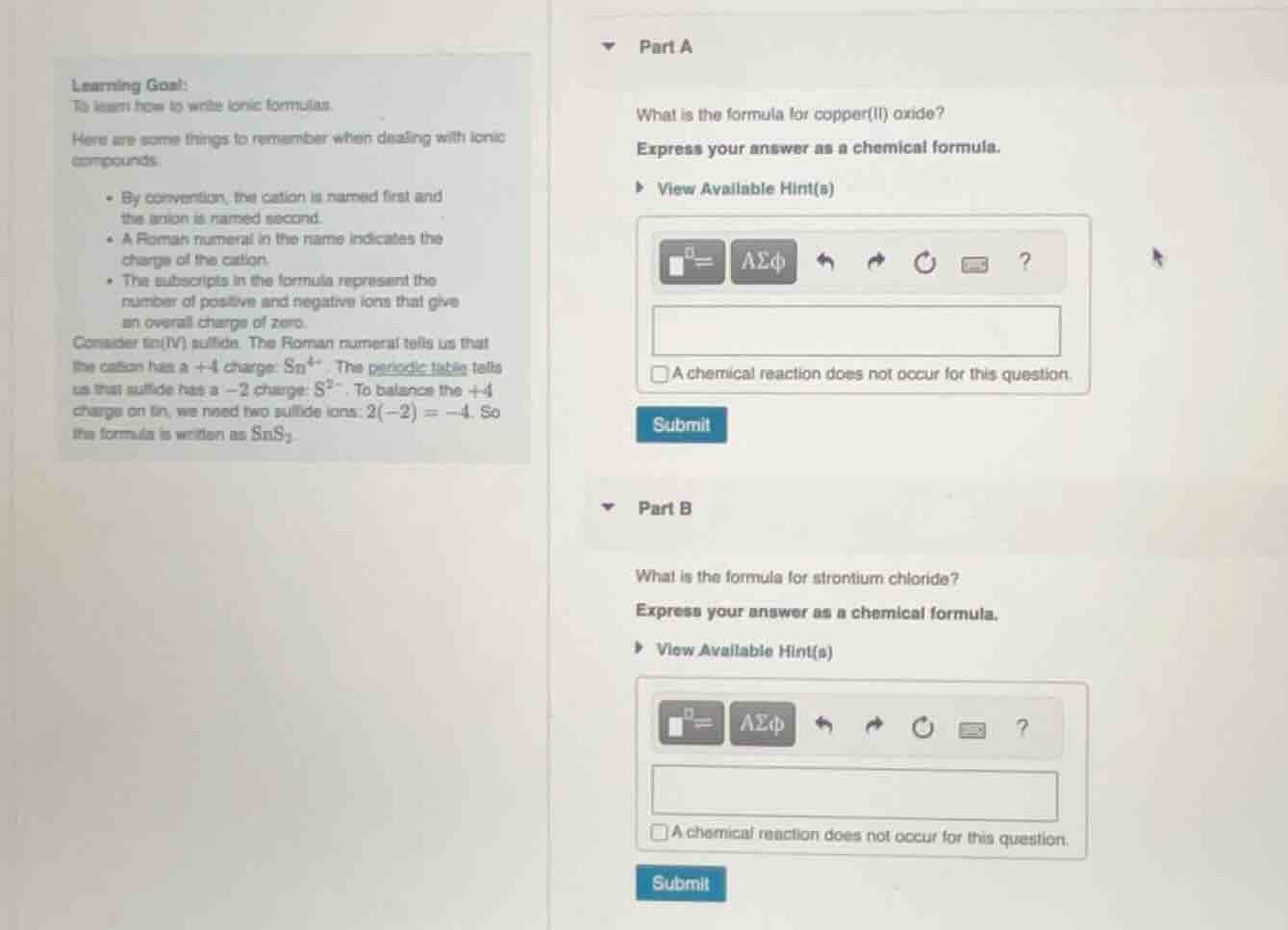
learning goal:
to learn how to write ionic formulas.
here are some things to remember when dealing with ionic compounds:
- by convention, the cation is named first and the anion is named second.
- a roman numeral in the name indicates the charge of the cation.
- the subscripts in the formula represent the number of positive and negative ions that give an overall charge of zero.
consider tin(iv) sulfide. the roman numeral tells us that the cation has a +4 charge: sn⁴⁺. the periodic table tells us that sulfide has a −2 charge: s²⁻. to balance the +4 charge on tin, we need two sulfide ions: 2(−2) = −4. so the formula is written as sns₂.
part a
what is the formula for copper(ii) oxide?
express your answer as a chemical formula.
view available hint(s)
a chemical reaction does not occur for this question.
submit
part b
what is the formula for strontium chloride?
express your answer as a chemical formula.
view available hint(s)
a chemical reaction does not occur for this question.
submit
Part A
Step1: Determine ion charges
Copper(II) has charge \( \text{Cu}^{2+} \), oxide is \( \text{O}^{2-} \).
Step2: Balance charges
To balance \( +2 \) (Cu) and \( -2 \) (O), we need 1 Cu and 1 O.
Step1: Determine ion charges
Strontium (group 2) has \( \text{Sr}^{2+} \), chloride is \( \text{Cl}^- \).
Step2: Balance charges
To balance \( +2 \) (Sr) and \( -1 \) (Cl), we need 1 Sr and 2 Cl.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( \text{CuO} \)