QUESTION IMAGE
Question
level: 3 challenge 5 of 5 score: 5 start over
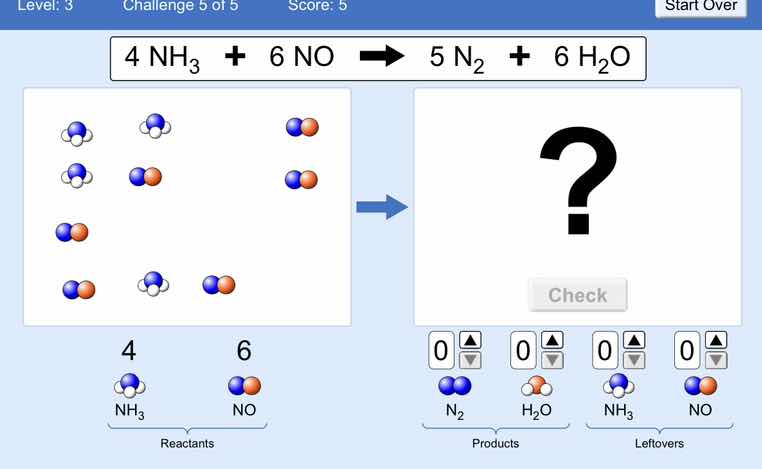
$4\\ nh_3 + 6\\ no \
ightarrow 5\\ n_2 + 6\\ h_2o$
4 $nh_3$
6 $no$
reactants
$n_2$ $h_2o$ $nh_3$ $no$
products leftovers
check
Step1: Count initial reactants
Initial: $4$ $\text{NH}_3$, $6$ $\text{NO}$
Step2: Match reaction stoichiometry
Reaction: $4\text{NH}_3 + 6\text{NO}
ightarrow 5\text{N}_2 + 6\text{H}_2\text{O}$
Reactants are used completely.
Step3: Calculate product amounts
$\text{N}_2$: $5$, $\text{H}_2\text{O}$: $6$; Leftovers: $0$ for all.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\text{N}_2$: 5
- $\text{H}_2\text{O}$: 6
- Leftover $\text{NH}_3$: 0
- Leftover $\text{NO}$: 0
The product box should contain 5 $\text{N}_2$ (blue-blue molecules) and 6 $\text{H}_2\text{O}$ (blue-white molecules), with no leftover reactants.