QUESTION IMAGE
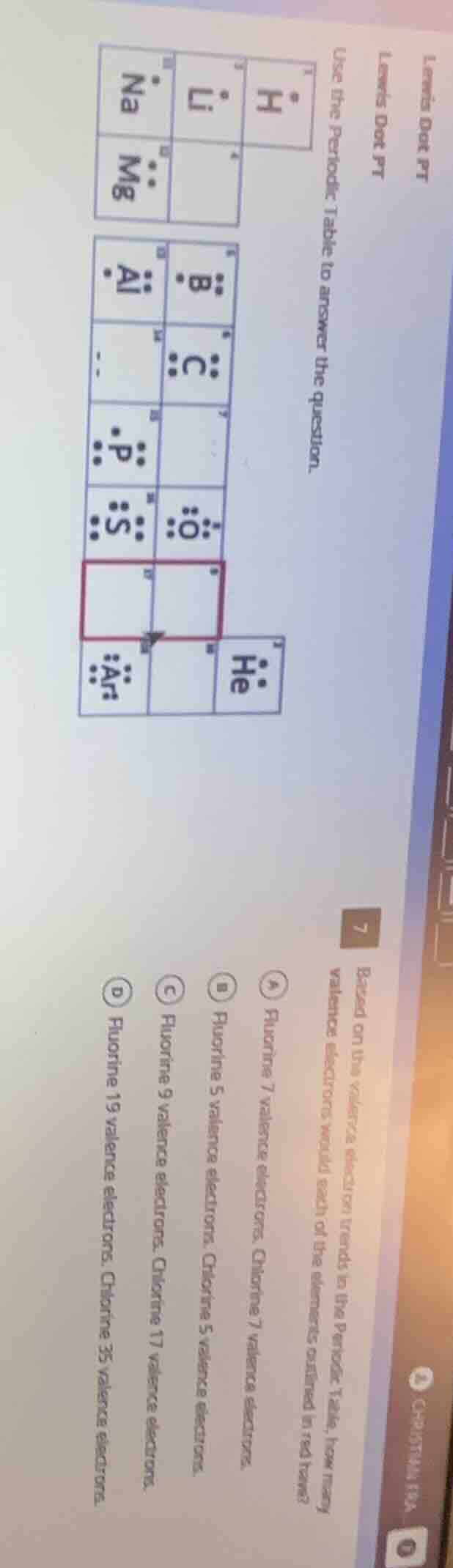
Question
lewis dot pt
lewis dot pt
use the periodic table to answer the question.
based on the valence electron trends in the periodic table, how many valence electrons would each of the elements outlined in red have?
a) fluorine 7 valence electrons, chlorine 7 valence electrons.
b) fluorine 5 valence electrons, chlorine 5 valence electrons.
c) fluorine 9 valence electrons, chlorine 17 valence electrons.
d) fluorine 19 valence electrons, chlorine 35 valence electrons.
The element in the red - outlined box is in Group 17 of the periodic table (halogens), along with fluorine (F) and chlorine (Cl). Elements in Group 17 have 7 valence electrons. Fluorine (atomic number 9) and chlorine (atomic number 17) both belong to Group 17, so they each have 7 valence electrons. Option A states "Fluorine 7 valence electrons, Chlorine 7 valence electrons", which is correct. Option B says 5 valence electrons, which is wrong. Option C confuses atomic number with valence electrons. Option D also confuses atomic mass (or atomic number - related misinterpretation) with valence electrons.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. Fluorine 7 valence electrons, Chlorine 7 valence electrons