QUESTION IMAGE
Question
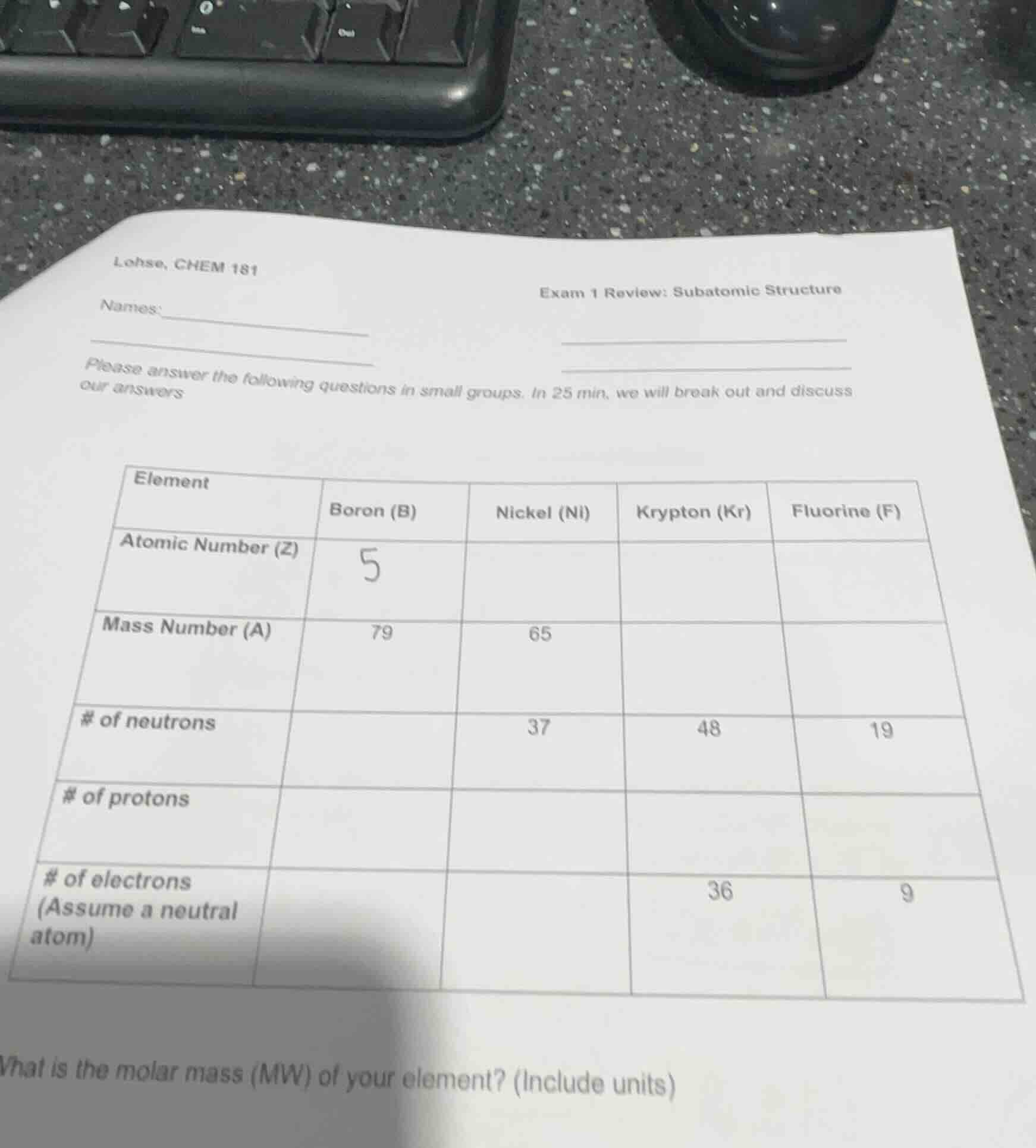
lohse, chem 181
exam 1 review: subatomic structure
names:
please answer the following questions in small groups. in 25 min, we will break out and discuss our answers
element | boron (b) | nickel (ni) | krypton (kr) | fluorine (f)
atomic number (z) | 5 | | |
mass number (a) | 79 | 65 | |
of neutrons | | 37 | 48 | 19
of protons | | | |
of electrons (assume a neutral atom) | | | 36 | 9
what is the molar mass (mw) of your element? (include units)
Step1: Recall key atomic relationships
For neutral atoms:
- Atomic Number (Z) = # of protons = # of electrons
- Mass Number (A) = # of protons + # of neutrons, so # of neutrons = A - Z
Step2: Fill in Boron's missing values
Substep2a: Calculate # of protons
Atomic Number (Z) = 5, so # of protons = 5
Substep2b: Calculate # of neutrons
$\text{# of neutrons} = A - Z = 79 - 5 = 74$
Substep2c: Calculate # of electrons (neutral atom)
of electrons = # of protons = 5
Step3: Fill in Nickel's missing values
Substep3a: Calculate # of protons
$\text{# of protons} = A - \text{# of neutrons} = 65 - 37 = 28$
Substep3b: Identify Atomic Number (Z)
Atomic Number (Z) = # of protons = 28
Substep3c: Calculate # of electrons (neutral atom)
of electrons = # of protons = 28
Step4: Fill in Krypton's missing values
Substep4a: Identify # of protons
of protons = # of electrons (neutral) = 36
Substep4b: Identify Atomic Number (Z)
Atomic Number (Z) = # of protons = 36
Substep4c: Calculate Mass Number (A)
$A = \text{# of protons} + \text{# of neutrons} = 36 + 48 = 84$
Step5: Fill in Fluorine's missing values
Substep5a: Identify # of protons
of protons = # of electrons (neutral) = 9
Substep5b: Identify Atomic Number (Z)
Atomic Number (Z) = # of protons = 9
Substep5c: Calculate Mass Number (A)
$A = \text{# of protons} + \text{# of neutrons} = 9 + 19 = 28$
Step6: Determine molar masses
Molar mass of an element (for the given isotope) equals its mass number, with units g/mol.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Completed Table:
| Element | Boron (B) | Nickel (Ni) | Krypton (Kr) | Fluorine (F) |
|---|---|---|---|---|
| Mass Number (A) | 79 | 65 | 84 | 28 |
| # of neutrons | 74 | 37 | 48 | 19 |
| # of protons | 5 | 28 | 36 | 9 |
| # of electrons (neutral atom) | 5 | 28 | 36 | 9 |
Molar Masses:
- Boron (given isotope): 79 g/mol
- Nickel (given isotope): 65 g/mol
- Krypton (given isotope): 84 g/mol
- Fluorine (given isotope): 28 g/mol