QUESTION IMAGE
Question
loudon | parise seventh edition
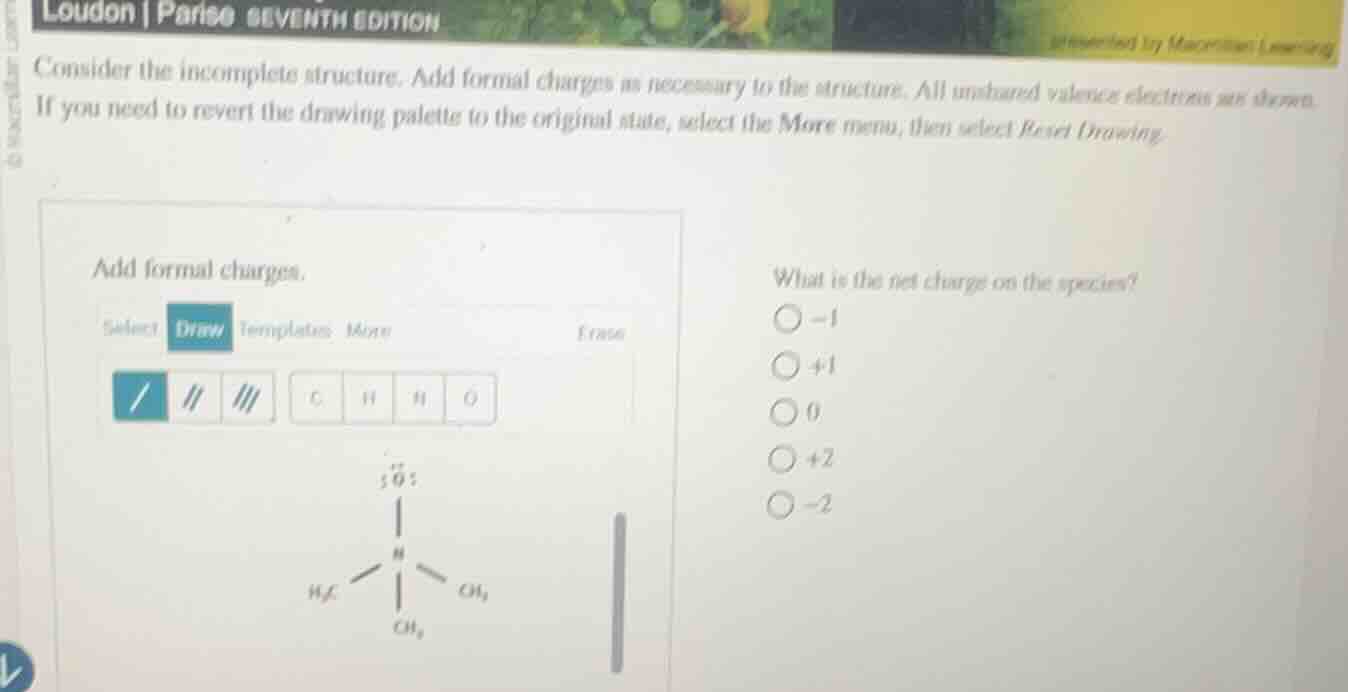
consider the incomplete structure. add formal charges as necessary to the structure. all unshared valence electrons are shown.
if you need to revert the drawing palettes to the original state, select the more menu, then select reset drawing.
add formal charges.
select draw template more erase
/ || |||
c h h o
what is the net charge on the species?
-1
+1
0
+2
-2
Step1: Calculate O formal charge
Formal charge formula: $FC = V - N - \frac{B}{2}$, where $V$=valence electrons, $N$=nonbonding electrons, $B$=bonding electrons.
For O: $V=6$, $N=6$, $B=2$.
$FC = 6 - 6 - \frac{2}{2} = -1$
Step2: Calculate C formal charges
For each $CH_3$ C and central C: $V=4$, $N=0$, $B=8$.
$FC = 4 - 0 - \frac{8}{2} = 0$
Step3: Calculate H formal charges
For all H: $V=1$, $N=0$, $B=2$.
$FC = 1 - 0 - \frac{2}{2} = 0$
Step4: Sum all formal charges
Net charge = $(-1) + 0+0+0+0+0+0+0 = -1$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
-1 (corresponding to the option $\boldsymbol{-1}$)
The formal charge on the oxygen atom is $-1$, and all other atoms have a formal charge of $0$.