QUESTION IMAGE
Question
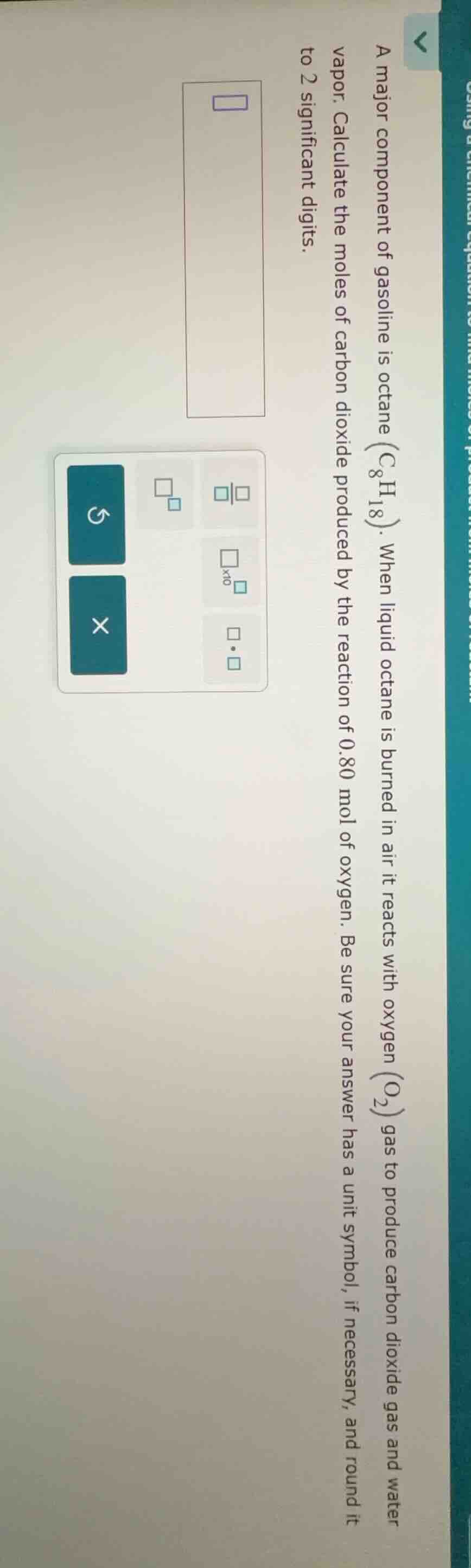
a major component of gasoline is octane ($ce{c_{8}h_{18}}$). when liquid octane is burned in air it reacts with oxygen ($ce{o_{2}}$) gas to produce carbon dioxide gas and water vapor. calculate the moles of carbon dioxide produced by the reaction of 0.80 mol of oxygen. be sure your answer has a unit symbol, if necessary, and round it to 2 significant digits.
Step1: Write the balanced equation
The combustion reaction of octane (\(C_8H_{18}\)) is:
Step2: Determine the mole ratio
From the balanced equation, the mole ratio of \(O_2\) to \(CO_2\) is \(\frac{25\ \text{mol}\ O_2}{16\ \text{mol}\ CO_2}\) (or inverted for calculation). We need moles of \(CO_2\) from moles of \(O_2\), so the ratio is \(\frac{16\ \text{mol}\ CO_2}{25\ \text{mol}\ O_2}\).
Step3: Calculate moles of \(CO_2\)
Given moles of \(O_2 = 0.80\ \text{mol}\).
Moles of \(CO_2 = 0.80\ \text{mol}\ O_2 \times \frac{16\ \text{mol}\ CO_2}{25\ \text{mol}\ O_2}\)
Simplify: \(0.80 \times \frac{16}{25} = 0.80 \times 0.64 = 0.512\ \text{mol}\)
Step4: Round to 2 significant digits
\(0.512\ \text{mol}\) rounded to 2 significant digits is \(0.51\ \text{mol}\) (or \(5.1 \times 10^{-1}\ \text{mol}\), but \(0.51\ \text{mol}\) is clearer). Wait, correction: \(0.80\) has 2 significant digits, so the calculation:
\(0.80 \times \frac{16}{25} = \frac{12.8}{25} = 0.512\). Rounded to 2 sig figs: \(0.51\ \text{mol}\)? Wait, no—\(0.80\) is two sig figs, \(16/25 = 0.64\) (exact ratio, so sig figs from \(0.80\)). \(0.80 \times 0.64 = 0.512\), which rounds to \(0.51\) (two sig figs: the first two non - zero digits? Wait, \(0.512\) rounded to two significant digits: the first two significant digits are 5 and 1, the next digit is 2 (less than 5), so it stays \(0.51\)? Wait, no—wait, \(0.80\) is two sig figs, so the result should have two sig figs. \(0.80 \times 16 / 25 = (0.80 \times 16)/25 = 12.8 / 25 = 0.512\). Rounded to two significant digits: look at the third digit (2), which is less than 5, so we keep the first two: \(0.51\) mol? Wait, no, \(0.512\) to two sig figs is \(0.51\) (since the first two sig figs are 5 and 1, the third is 2, so we don't round up). Wait, but let's check the calculation again. \(16/25 = 0.64\), \(0.80 \times 0.64 = 0.512\). Yes, so two sig figs: \(0.51\ \text{mol}\) (or \(5.1 \times 10^{-1}\ \text{mol}\)). Wait, but maybe I made a mistake in the ratio? Let's re - check the balanced equation: 2 moles of octane react with 25 moles of \(O_2\) to produce 16 moles of \(CO_2\). So moles of \(CO_2\) = (moles of \(O_2\) × 16) / 25. So 0.80 × 16 = 12.8; 12.8 / 25 = 0.512. Rounded to two significant figures: 0.51 mol (or 5.1 × 10⁻¹ mol). But 0.51 has two significant digits (5 and 1). Alternatively, maybe the answer is 0.51 mol, or 5.1×10⁻¹ mol. Wait, but let's confirm: 0.80 is two sig figs, so the answer should have two. So 0.51 mol (or 0.51, which is two sig figs).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(0.51\ \text{mol}\) (or \(5.1 \times 10^{-1}\ \text{mol}\), but \(0.51\ \text{mol}\) is more direct)