QUESTION IMAGE
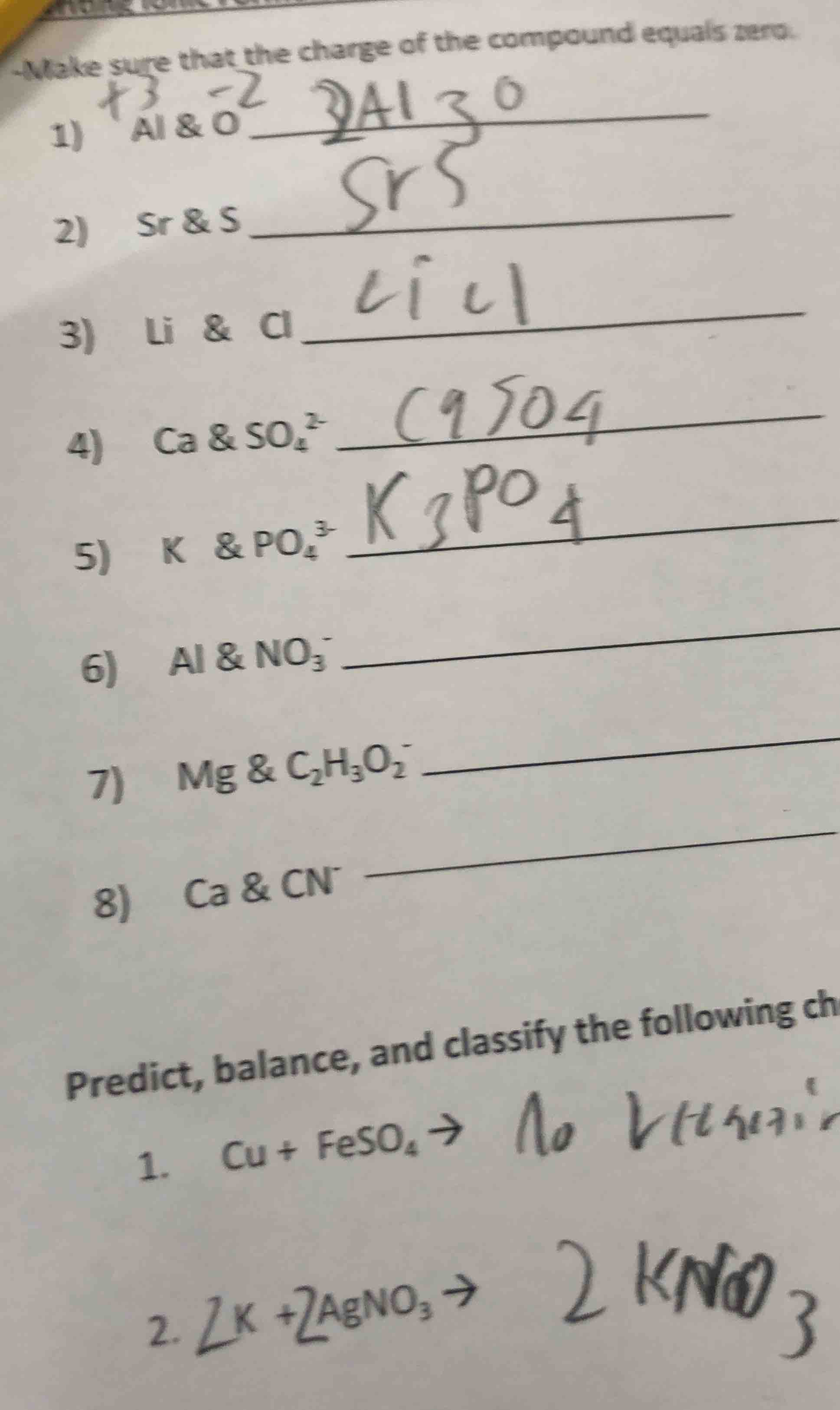
Question
-make sure that the change of the compound equals zero.
- $\stackrel{+3}{\text{al}}$ & $\stackrel{-2}{\text{o}}$
- sr & s
- li & cl
- ca & $\text{so}_4^{2-}$
- k & $\text{po}_4^{3-}$
- al & $\text{no}_3^-$
- mg & $\text{c}_2\text{h}_3\text{o}_2^-$
- ca & $\text{cn}^-$
predict, balance, and classify the following ch
- cu + $\text{feso}_4$ $\
ightarrow$
- $2\text{k}$ + $2\text{agno}_3$ $\
ightarrow$
Step1: Fix Al & O formula
Al has +3 charge, O has -2 charge. Use crisscross method: $\text{Al}_2\text{O}_3$
Step2: Fix Sr & S formula
Sr has +2 charge, S has -2 charge. Simplify ratio: $\text{SrS}$
Step3: Confirm Li & Cl formula
Li has +1 charge, Cl has -1 charge: $\text{LiCl}$
Step4: Fix Ca & $\text{SO}_4^{2-}$ formula
Ca has +2 charge, $\text{SO}_4^{2-}$ has -2 charge: $\text{CaSO}_4$
Step5: Confirm K & $\text{PO}_4^{3-}$ formula
K has +1 charge, $\text{PO}_4^{3-}$ has -3 charge: $\text{K}_3\text{PO}_4$
Step6: Find Al & $\text{NO}_3^-$ formula
Al has +3 charge, $\text{NO}_3^-$ has -1 charge. Crisscross: $\text{Al(NO}_3\text{)}_3$
Step7: Find Mg & $\text{C}_2\text{H}_3\text{O}_2^-$ formula
Mg has +2 charge, $\text{C}_2\text{H}_3\text{O}_2^-$ has -1 charge. Crisscross: $\text{Mg(C}_2\text{H}_3\text{O}_2\text{)}_2$
Step8: Find Ca & $\text{CN}^-$ formula
Ca has +2 charge, $\text{CN}^-$ has -1 charge. Crisscross: $\text{Ca(CN)}_2$
Step9: Analyze Cu + $\text{FeSO}_4$ reaction
Cu is less reactive than Fe, so no reaction: $\text{Cu} + \text{FeSO}_4
ightarrow \text{No Reaction}$
Step10: Balance K + $\text{AgNO}_3$ reaction
K has +1, $\text{AgNO}_3$ has $\text{Ag}^+$; products are $\text{KNO}_3$ and Ag. Balanced: $2\text{K} + 2\text{AgNO}_3
ightarrow 2\text{KNO}_3 + 2\text{Ag}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $\text{Al}_2\text{O}_3$
- $\text{SrS}$
- $\text{LiCl}$
- $\text{CaSO}_4$
- $\text{K}_3\text{PO}_4$
- $\text{Al(NO}_3\text{)}_3$
- $\text{Mg(C}_2\text{H}_3\text{O}_2\text{)}_2$
- $\text{Ca(CN)}_2$
- $\text{Cu} + \text{FeSO}_4
ightarrow \text{No Reaction}$ (single displacement, non-spontaneous)
- $2\text{K} + 2\text{AgNO}_3
ightarrow 2\text{KNO}_3 + 2\text{Ag}$ (single displacement, spontaneous)