QUESTION IMAGE
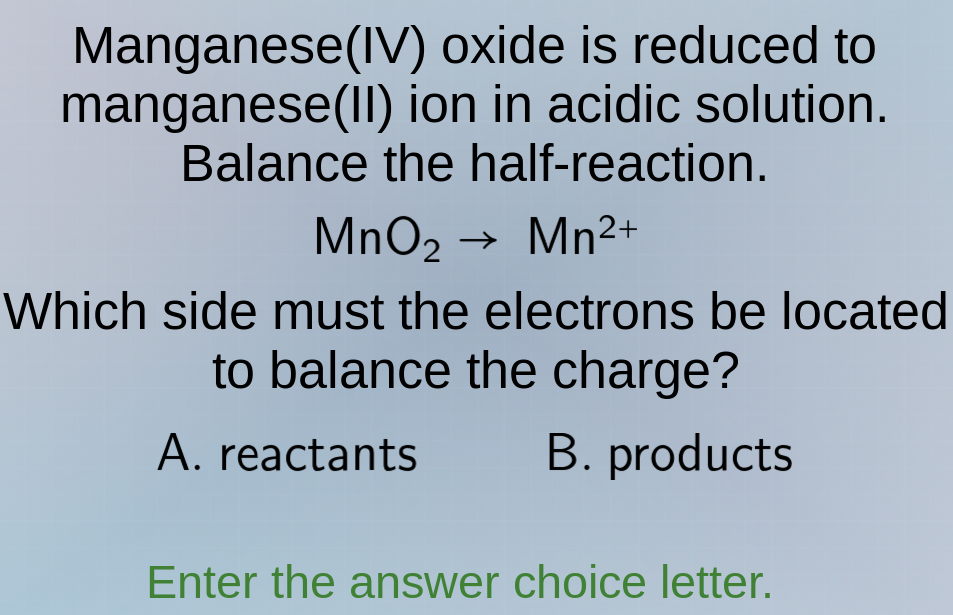
Question
manganese(iv) oxide is reduced to manganese(ii) ion in acidic solution. balance the half-reaction. mno₂ → mn²⁺ which side must the electrons be located to balance the charge? a. reactants b. products enter the answer choice letter.
- First, balance the Mn atoms: \( \text{MnO}_2
ightarrow \text{Mn}^{2+} \) (Mn is already balanced, 1 on each side).
- Balance O by adding \( \text{H}_2\text{O} \): \( \text{MnO}_2
ightarrow \text{Mn}^{2+} + 2\text{H}_2\text{O} \) (2 O on left, so 2 \( \text{H}_2\text{O} \) on right).
- Balance H by adding \( \text{H}^+ \) (acidic solution): \( \text{MnO}_2 + 4\text{H}^+
ightarrow \text{Mn}^{2+} + 2\text{H}_2\text{O} \) (4 H on left from 4 \( \text{H}^+ \), 4 H on right from 2 \( \text{H}_2\text{O} \)).
- Balance charge: Left side charge: \( \text{MnO}_2 \) (neutral) + \( 4\text{H}^+ \) (4+ charge) = 4+ total. Right side: \( \text{Mn}^{2+} \) (2+ charge) + \( 2\text{H}_2\text{O} \) (neutral) = 2+ total. The difference is 2 electrons (since reduction means gain of electrons, and we need to add electrons to the left (reactants) to balance charge? Wait, no: Wait, the left has 4+ (from \( 4\text{H}^+ \)), right has 2+ (from \( \text{Mn}^{2+} \)). To balance charge, we need to add electrons to the left (reactants) to reduce the positive charge? Wait, no: Wait, reduction is gain of electrons. The Mn in \( \text{MnO}_2 \) has oxidation state +4, and in \( \text{Mn}^{2+} \) it's +2, so it's gaining 2 electrons (reduction). So the half - reaction with electrons: \( \text{MnO}_2 + 4\text{H}^+ + 2e^-
ightarrow \text{Mn}^{2+} + 2\text{H}_2\text{O} \). So electrons are on the reactants side (left side). So the answer is A.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A