QUESTION IMAGE
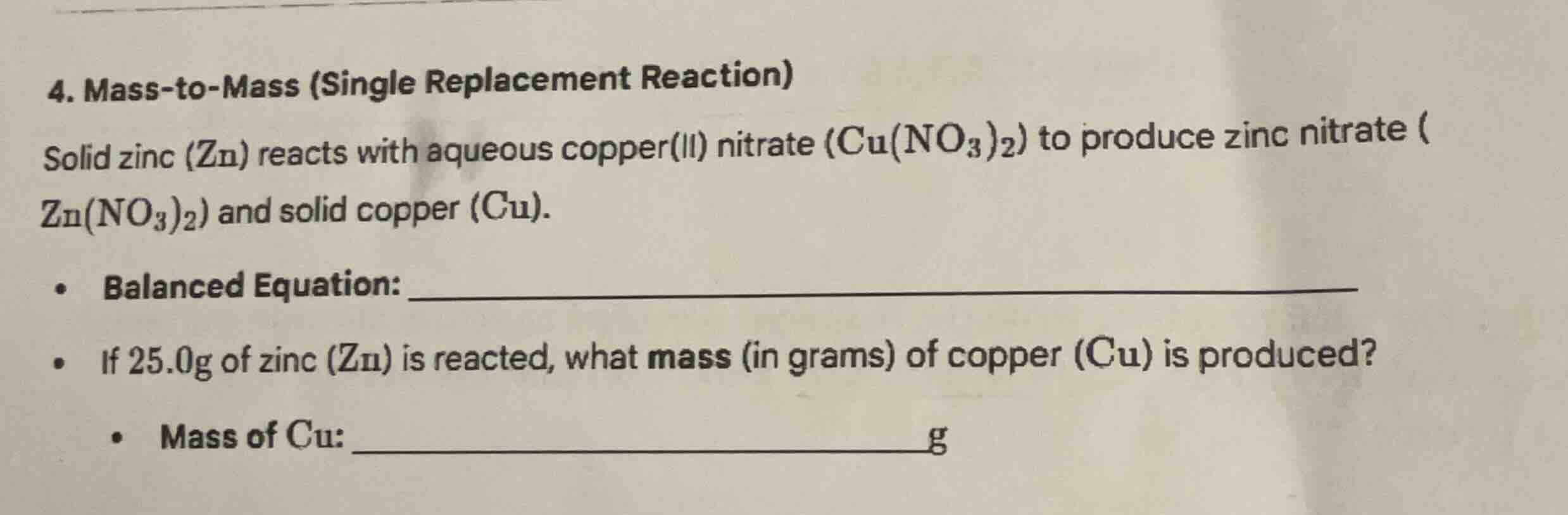
Question
- mass-to-mass (single replacement reaction)
solid zinc (zn) reacts with aqueous copper(ii) nitrate ($\text{cu(no}_3\text{)}_2$) to produce zinc nitrate ($\text{zn(no}_3\text{)}_2$) and solid copper (cu).
- balanced equation: ____________
- if 25.0g of zinc (zn) is reacted, what mass (in grams) of copper (cu) is produced?
- mass of cu: ____________g
Step1: Balance the chemical equation
$\text{Zn}(s) + \text{Cu(NO}_3\text{)}_2(aq)
ightarrow \text{Zn(NO}_3\text{)}_2(aq) + \text{Cu}(s)$
Step2: Find molar masses
Molar mass of $\text{Zn} = 65.38\ \text{g/mol}$, Molar mass of $\text{Cu} = 63.55\ \text{g/mol}$
Step3: Calculate moles of Zn
$\text{Moles of Zn} = \frac{25.0\ \text{g}}{65.38\ \text{g/mol}} \approx 0.3824\ \text{mol}$
Step4: Relate moles Zn to Cu
From balanced equation, 1 mol Zn produces 1 mol Cu, so moles of Cu = 0.3824 mol
Step5: Calculate mass of Cu
$\text{Mass of Cu} = 0.3824\ \text{mol} \times 63.55\ \text{g/mol} \approx 24.3\ \text{g}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Balanced Equation: $\boldsymbol{\text{Zn}(s) + \text{Cu(NO}_3\text{)}_2(aq)
ightarrow \text{Zn(NO}_3\text{)}_2(aq) + \text{Cu}(s)}$
Mass of Cu: $\boldsymbol{24.3}$ g