QUESTION IMAGE
Question
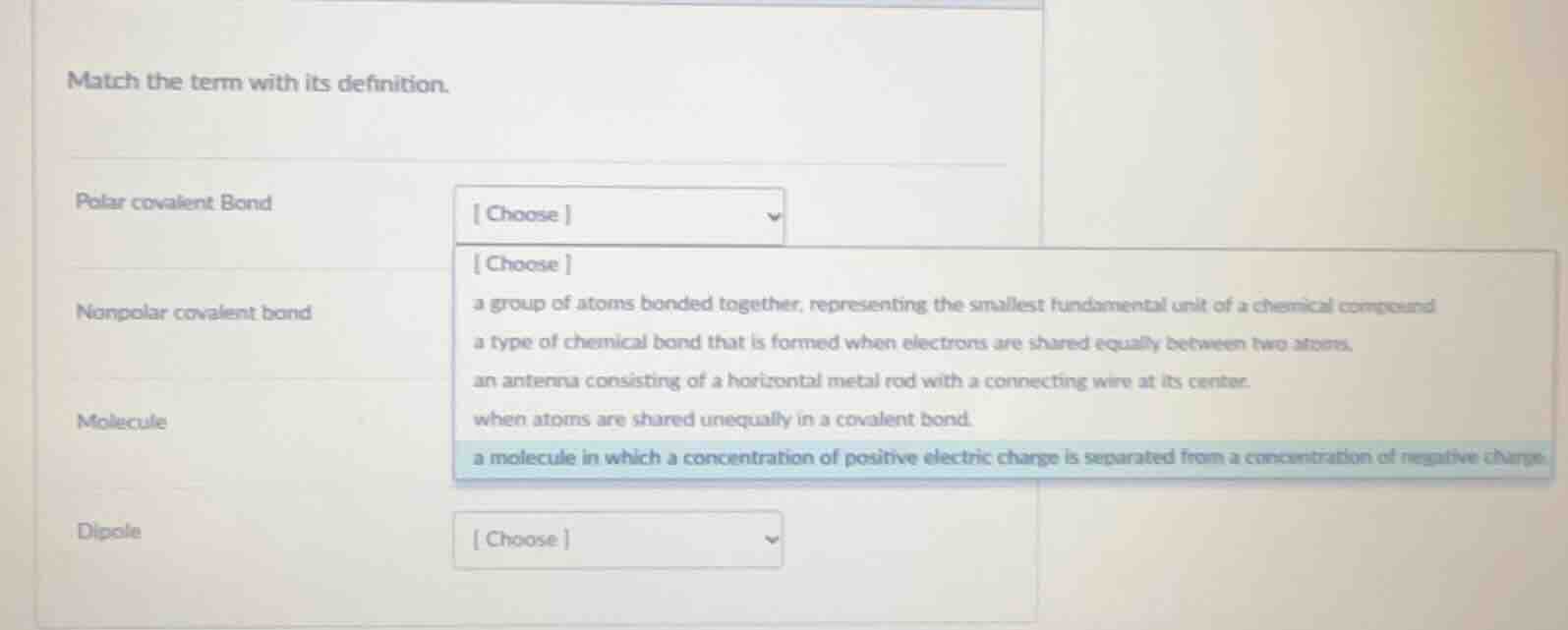
match the term with its definition.
polar covalent bond choose
choose
nonpolar covalent bond a group of atoms bonded together, representing the smallest fundamental unit of a chemical compound
a type of chemical bond that is formed when electrons are shared equally between two atoms.
an antenna consisting of a horizontal metal rod with a connecting wire at its center.
when atoms are shared unequally in a covalent bond.
a molecule in which a concentration of positive electric charge is separated from a concentration of negative charge.
molecule
dipole choose
- Polar covalent Bond: Defined by unequal sharing of electrons in a covalent bond. So matches "when atoms are shared unequally in a covalent bond".
- Nonpolar covalent bond: Formed when electrons are shared equally between two atoms, so matches "a type of chemical bond that is formed when electrons are shared equally between two atoms".
- Molecule: A group of atoms bonded together, representing the smallest fundamental unit of a chemical compound, so matches that definition.
- Dipole: A molecule in which a concentration of positive electric charge is separated from a concentration of negative charge, so matches that definition.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Polar covalent Bond: when atoms are shared unequally in a covalent bond
- Nonpolar covalent bond: a type of chemical bond that is formed when electrons are shared equally between two atoms
- Molecule: a group of atoms bonded together, representing the smallest fundamental unit of a chemical compound
- Dipole: a molecule in which a concentration of positive electric charge is separated from a concentration of negative charge