QUESTION IMAGE
Question
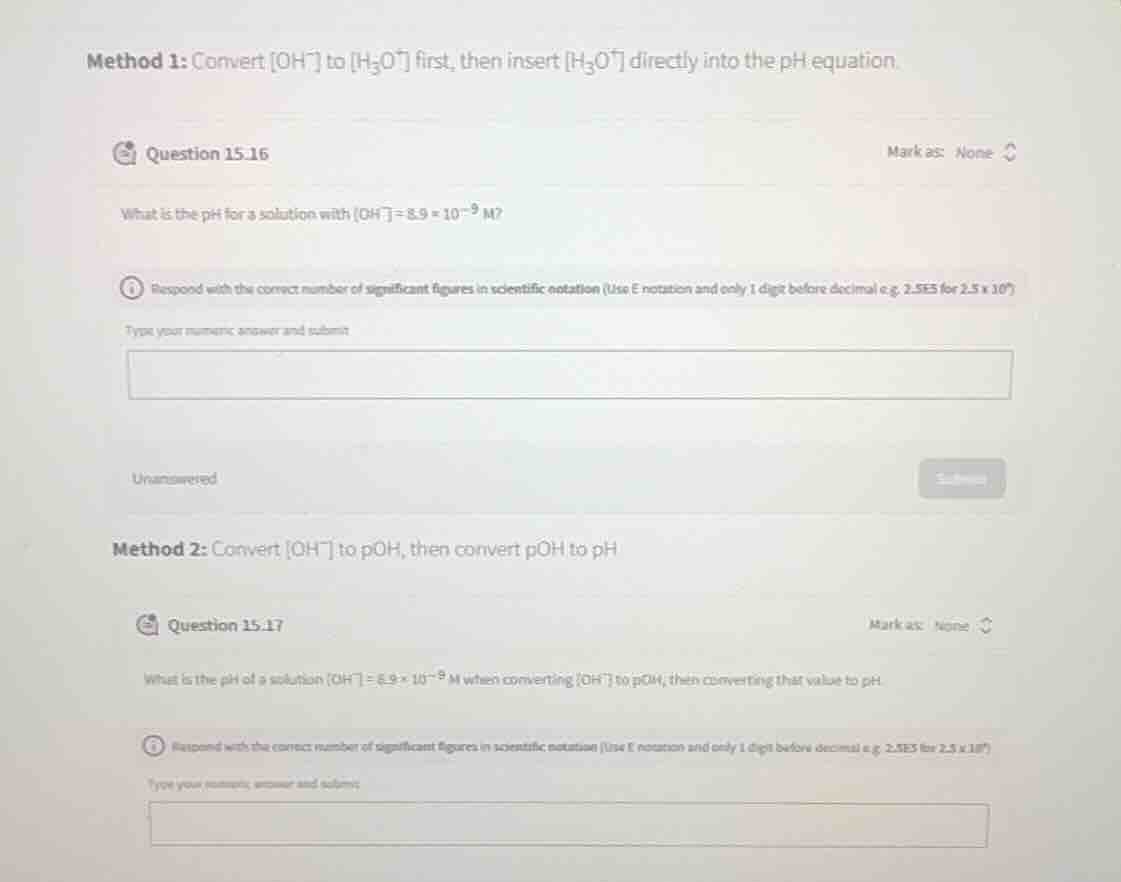
method 1: convert $oh^-$ to $h_3o^+$ first, then insert $h_3o^+$ directly into the ph equation.
question 15.16
mark as: none
what is the ph for a solution with $oh^-=8.9 \times 10^{-9}$ m?
respond with the correct number of significant figures in scientific notation (use e notation and only 1 digit before decimal e.g. 2.5e5 for $2.5 \times 10^5$)
type your numeric answer and submit
unanswered
submit
method 2: convert $oh^-$ to poh, then convert poh to ph
question 15.17
mark as: none
what is the ph of a solution $oh^- = 8.9 \times 10^{-9}$ m when converting $oh^-$ to poh, then converting that value to ph
respond with the correct number of significant figures in scientific notation (use e notation and only 1 digit before decimal e.g. 2.5e5 for $2.5 \times 10^5$)
type your numeric answer and submit
Step1: Use ion product of water
At 25°C, $K_w = [\text{H}_3\text{O}^+][\text{OH}^-] = 1.0 \times 10^{-14}$. Rearrange to solve for $[\text{H}_3\text{O}^+]$:
$$[\text{H}_3\text{O}^+] = \frac{1.0 \times 10^{-14}}{[\text{OH}^-]}$$
Substitute $[\text{OH}^-] = 8.9 \times 10^{-9}\ \text{M}$:
$$[\text{H}_3\text{O}^+] = \frac{1.0 \times 10^{-14}}{8.9 \times 10^{-9}} \approx 1.124 \times 10^{-6}\ \text{M}$$
Step2: Calculate pH from $[\text{H}_3\text{O}^+]$
Use $pH = -\log_{10}[\text{H}_3\text{O}^+]$:
$$pH = -\log_{10}(1.124 \times 10^{-6}) \approx 5.95$$
Convert to required scientific notation: $6.0\text{E}0$ (2 significant figures, matching $8.9 \times 10^{-9}$)
---
Step1: Calculate pOH from $[\text{OH}^-]$
Use $p\text{OH} = -\log_{10}[\text{OH}^-]$, substitute $[\text{OH}^-] = 8.9 \times 10^{-9}\ \text{M}$:
$$p\text{OH} = -\log_{10}(8.9 \times 10^{-9}) \approx 8.05$$
Step2: Convert pOH to pH
Use $pH + p\text{OH} = 14.00$:
$$pH = 14.00 - 8.05 = 5.95$$
Convert to required scientific notation: $6.0\text{E}0$ (2 significant figures)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 15.16: $6.0\text{E}0$
Question 15.17: $6.0\text{E}0$