QUESTION IMAGE
Question
the modern atomic theory
quiz complete
1 2 3 4 5 6 7 8 9 10
time f
5
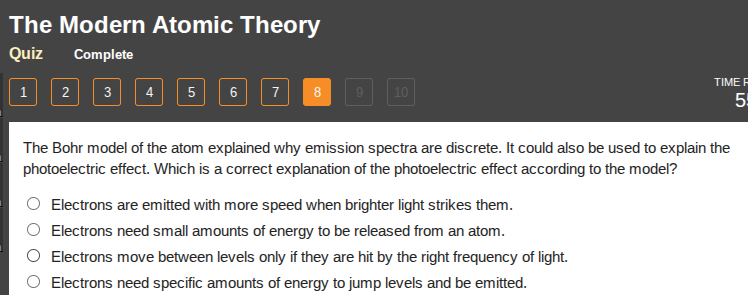
the bohr model of the atom explained why emission spectra are discrete. it could also be used to explain the photoelectric effect. which is a correct explanation of the photoelectric effect according to the model?
○ electrons are emitted with more speed when brighter light strikes them.
○ electrons need small amounts of energy to be released from an atom.
○ electrons move between levels only if they are hit by the right frequency of light.
○ electrons need specific amounts of energy to jump levels and be emitted.
atom (a specific amount). Brighter light only increases the number of photons, not electron speed; electrons need a minimum specific energy, not a small amount; and the option about frequency refers to absorption for level jumps, not the emission aspect of the photoelectric effect.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
atom (a specific amount). Brighter light only increases the number of photons, not electron speed; electrons need a minimum specific energy, not a small amount; and the option about frequency refers to absorption for level jumps, not the emission aspect of the photoelectric effect.