QUESTION IMAGE
Question
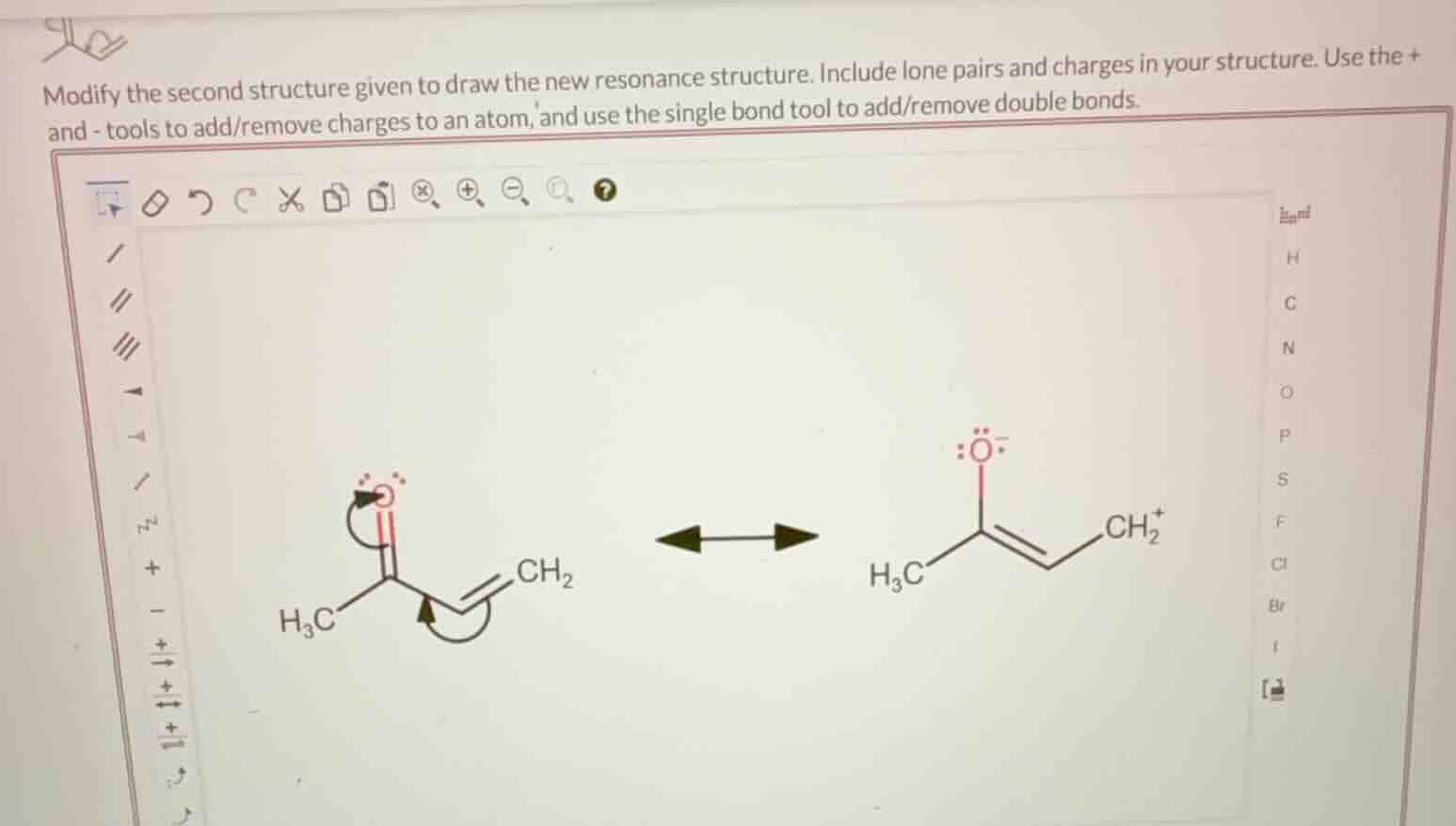
modify the second structure given to draw the new resonance structure. include lone pairs and charges in your structure. use the + and - tools to add/remove charges to an atom, and use the single bond tool to add/remove double bonds.
Step1: Analyze electron movement
The left structure shows electron movement: the lone pair on O moves to form a double bond, and the double bond in the alkene moves to form a carbocation. For the right structure, we reverse this. The double bond between the carbonyl - like carbon and the alkene carbon (in the right structure, the double bond is between the two carbons adjacent to the carbonyl - like carbon and \(CH_2^+\)) will move, and the lone pair on O (with a negative charge) will participate.
Step2: Modify the bonds and charges
- For the double bond between the carbon connected to \(H_3C\) and the adjacent carbon (in the right structure, the double bond is \(C = C\) near \(CH_2^+\)): we need to move the double bond. The \(CH_2^+\) will lose its positive charge as the double bond moves, and the oxygen will change its bond.
- The oxygen in the right structure has a negative charge and lone pairs. We will change the double bond between the carbonyl - like carbon (connected to \(H_3C\)) and the adjacent carbon. The double bond between the two carbons (the one with \(H_3C\) and the one in the double bond with \(CH_2^+\)) will be converted to a single bond, and the double bond in the alkene (between the carbon and \(CH_2^+\)) will be moved to form a double bond with the oxygen. Wait, no, let's correct. The resonance involves the movement of the pi electrons. In the left structure, the carbonyl group (C = O) has electrons moving, and the alkene (C = C) has electrons moving. In the right structure, to get the resonance structure, we need to move the double bond from the C = C (near \(CH_2^+\)) to form a double bond with the oxygen, and the \(CH_2^+\) will become neutral, and the oxygen will have a double bond (losing the negative charge? No, wait, the oxygen has a negative charge in the right structure. Let's draw the correct resonance.
The right structure is \(H_3C - C(=O^-) - C = CH_2^+\)? No, the right structure is \(H_3C - C(=O^-) - C = CH_2^+\) (with the negative charge on O and positive on \(CH_2\)). Wait, no, the right structure as given: \(H_3C - C(=O^-) - C = CH_2^+\) (the O has three lone pairs and a negative charge, the C - C is double, and \(CH_2\) is positive). To draw the resonance structure, we move the double bond from \(C = CH_2^+\) to form a double bond with the O, and the \(CH_2^+\) becomes \(CH_2\) (neutral), and the O loses its negative charge (since it forms a double bond, so O will have two lone pairs and a double bond, so charge 0? No, wait, the formal charge: O with a double bond (C = O) and two lone pairs has formal charge 0, and the carbon that was \(CH_2^+\) will have a positive charge? Wait, no, let's do formal charge.
Formal charge on O: in the right structure, O has 6 valence electrons (3 lone pairs: 6 electrons) and a single bond? No, the right structure has O with a double bond? Wait, the right structure shows O with a double bond? Wait, the right structure: \(H_3C - C(=O^-) - C = CH_2^+\). So O has a double bond (C = O) and a negative charge? No, if O is double - bonded, it has 2 lone pairs (4 electrons) and a double bond (2 electrons), total 6, formal charge = 6 - (4 + 2) = 0. But the right structure shows O with a negative charge, so maybe O is single - bonded. Wait, the left structure has O with a double bond (C = O) and a lone pair? No, the left structure's O has a double bond and a curved arrow, meaning electrons are moving.
Let's start over. The left structure: \(H_3C - C(=O) - C = CH_2\) (with arrows showing electron movement: one arrow from O's lone pair to form a double bond, and one…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The resonance structure is drawn by:
- Converting the double bond between the carbon adjacent to \(CH_2^+\) and \(CH_2^+\) to a single bond.
- Converting the single bond between the carbon (connected to \(H_3C\)) and oxygen to a double bond.
- Removing the positive charge from \(CH_2^+\) and the negative charge from O. The final structure will have \(H_3C - C(=O) - C = CH_2\) (with O having two lone pairs, double - bonded to C, and \(CH_2\) neutral, and the middle C - C double - bonded).