QUESTION IMAGE
Question
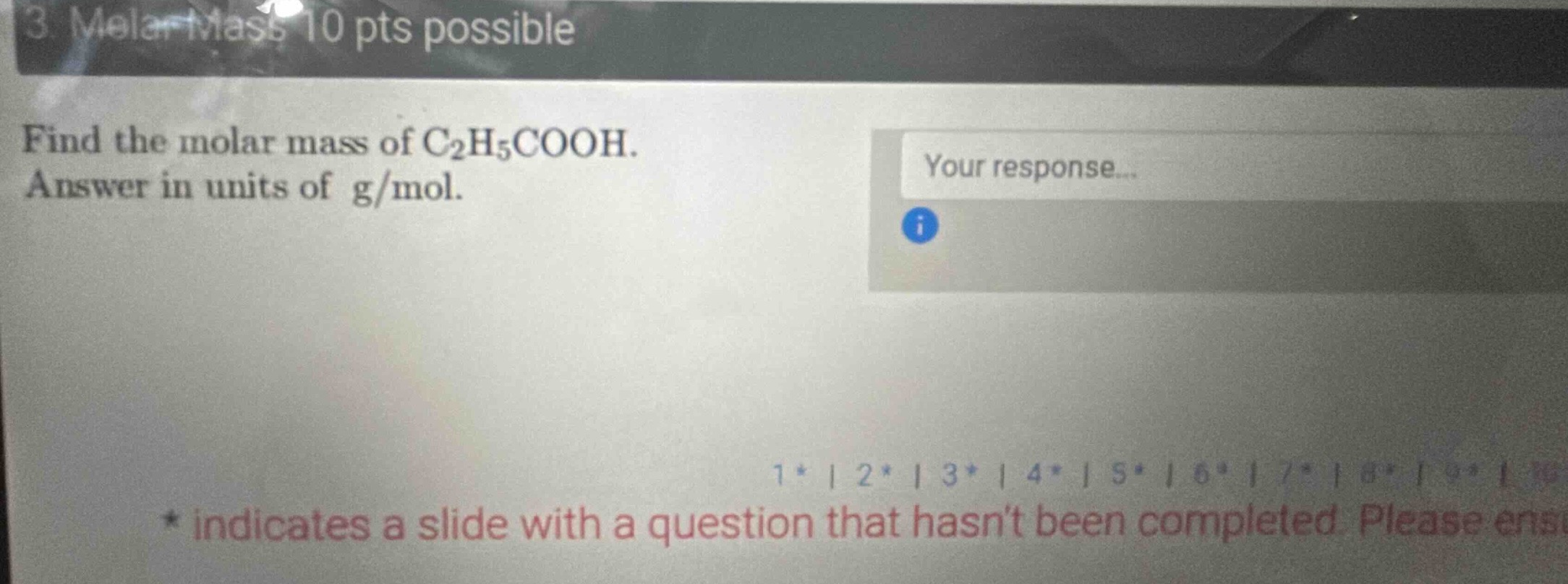
- molar mass 10 pts possible
find the molar mass of c₂h₅cooh.
answer in units of g/mol.
your response...
- indicates a slide with a question that hasnt been completed. please ensu
Step1: Identify atomic masses
Carbon (C): $12.01\ g/mol$, Hydrogen (H): $1.008\ g/mol$, Oxygen (O): $16.00\ g/mol$.
Step2: Count atoms in $\ce{C2H5COOH}$
- C: $2 + 1 = 3$ atoms (from $\ce{C2}$ and $\ce{COOH}$)
- H: $5 + 1 = 6$ atoms (from $\ce{C2H5}$ and $\ce{COOH}$)
- O: $2$ atoms (from $\ce{COOH}$)
Step3: Calculate molar mass
Molar mass = (3×12.01) + (6×1.008) + (2×16.00)
= $36.03 + 6.048 + 32.00$
= $74.078\ g/mol$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$74.08$ (rounded to two decimal places, or $74.078$)