QUESTION IMAGE
Question
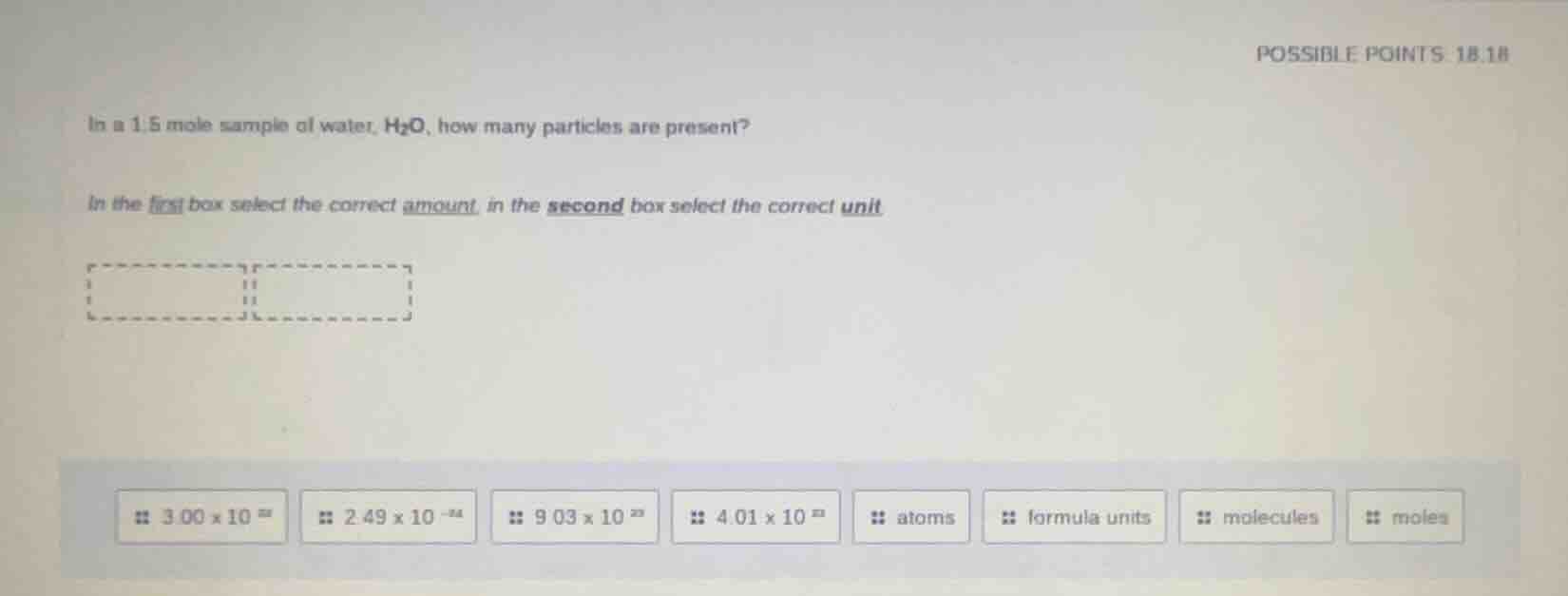
in a 1.5 mole sample of water, h₂o, how many particles are present?
in the first box select the correct amount, in the second box select the correct unit.
possible options for amount: 3.00×10²⁴, 2.49×10⁻²⁴, 9.03×10²³, 4.01×10²³
possible options for unit: atoms, formula units, molecules, moles
Step1: Recall Avogadro's number
Avogadro's number is \(6.02\times10^{23}\) particles per mole.
Step2: Calculate number of particles
To find the number of particles (molecules, since \(H_2O\) is a molecular compound) in 1.5 moles of \(H_2O\), we use the formula: Number of particles = moles \(\times\) Avogadro's number.
So, Number of molecules = \(1.5\space mol\times6.02\times10^{23}\space molecules/mol\)
\(1.5\times6.02 = 9.03\)
So the number of molecules is \(9.03\times10^{23}\) and the unit is molecules (since \(H_2O\) exists as molecules).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
First box: \(9.03\times10^{23}\)
Second box: molecules