QUESTION IMAGE
Question
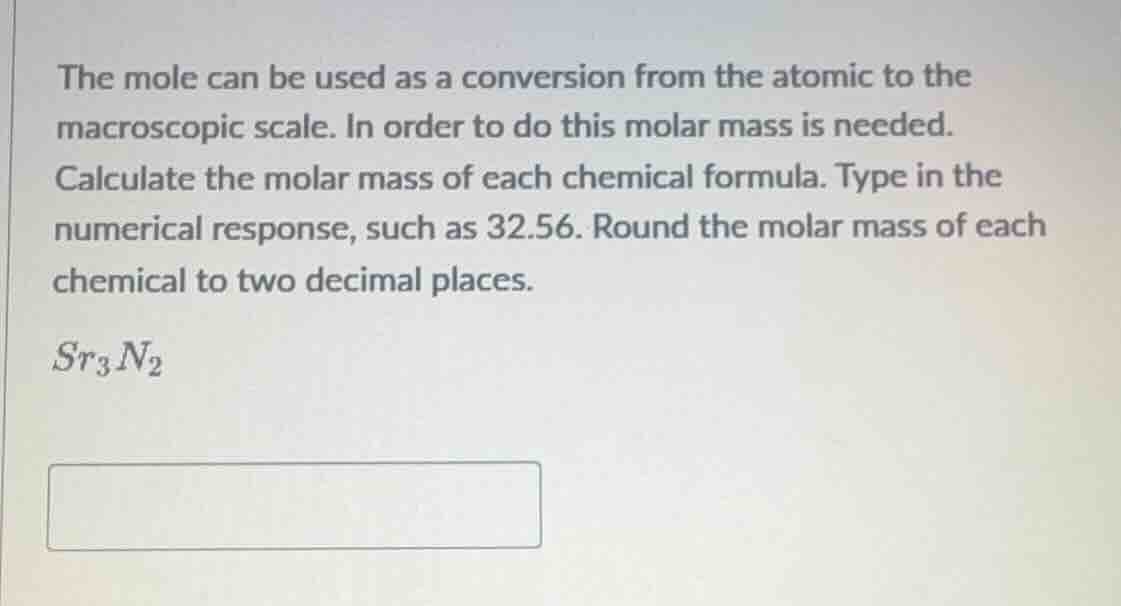
the mole can be used as a conversion from the atomic to the macroscopic scale. in order to do this molar mass is needed. calculate the molar mass of each chemical formula. type in the numerical response, such as 32.56. round the molar mass of each chemical to two decimal places. sr₃n₂
Step1: Find atomic masses
Atomic mass of Sr: $87.62\space g/mol$, atomic mass of N: $14.01\space g/mol$.
Step2: Calculate total mass for each element
For Sr: $3\times87.62 = 262.86\space g/mol$.
For N: $2\times14.01 = 28.02\space g/mol$.
Step3: Sum the masses
Molar mass of $Sr_3N_2$: $262.86 + 28.02 = 290.88\space g/mol$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
290.88