QUESTION IMAGE
Question
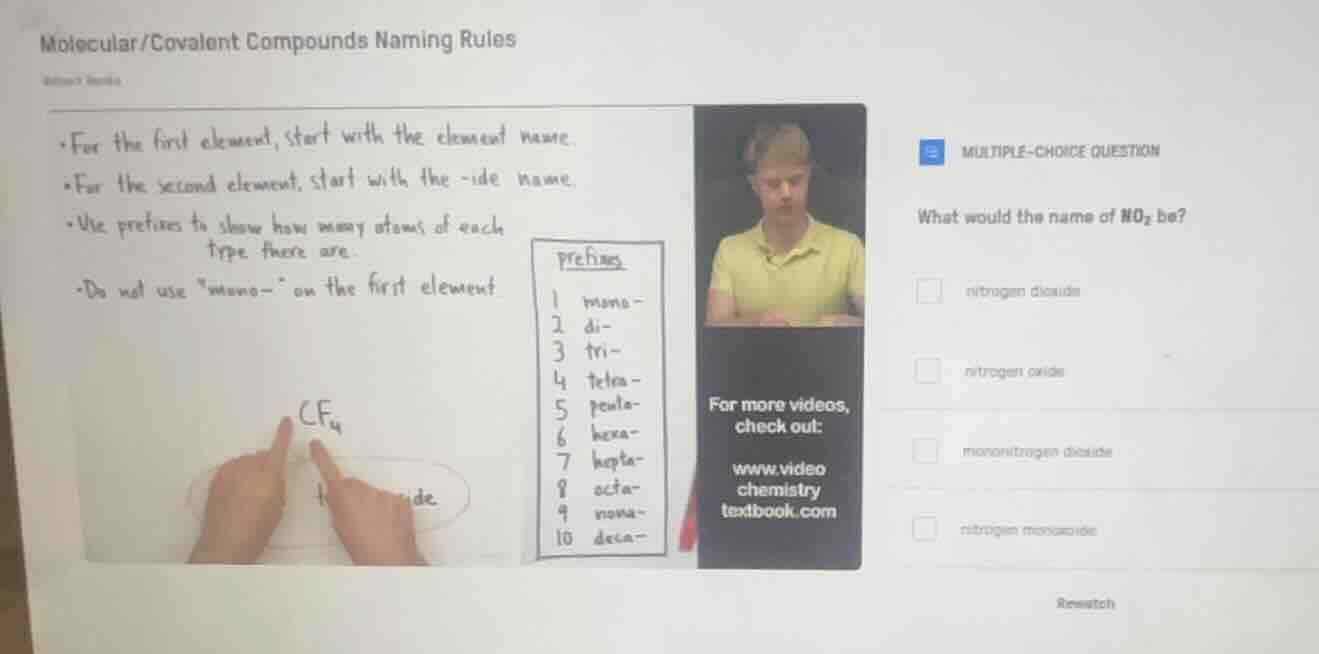
molecular/covalent compounds naming rules
- for the first element, start with the element name.
- for the second element, start with the -ide name.
- use prefixes to show how many atoms of each type there are.
- do not use \mono-\ on the first element
(with an example cf₄ and a prefixes table: 1 mono-, 2 di-, 3 tri-, 4 tetra-, 5 penta-, 6 hexa-, 7 hepta-, 8 octa-, 9 nona-, 10 deca-)
multiple-choice question
what would the name of no₂ be?
- nitrogen dioxide
- nitrogen oxide
- mononitrogen dioxide
- nitrogen monoxide
Step1: Analyze the first element in \( NO_2 \)
The first element is nitrogen (N). Since there is 1 atom of N, and we don't use "mono - " on the first element, we just use "nitrogen".
Step2: Analyze the second element in \( NO_2 \)
The second element is oxygen (O). The suffix for the second element in a covalent compound is "-ide", so we start with "oxide". Now, we look at the number of O atoms, which is 2. The prefix for 2 is "di - ", so we get "dioxide".
Step3: Combine the names of the two elements
Combining the name of the first element and the second element with the prefix, we get "nitrogen dioxide".
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
nitrogen dioxide