QUESTION IMAGE
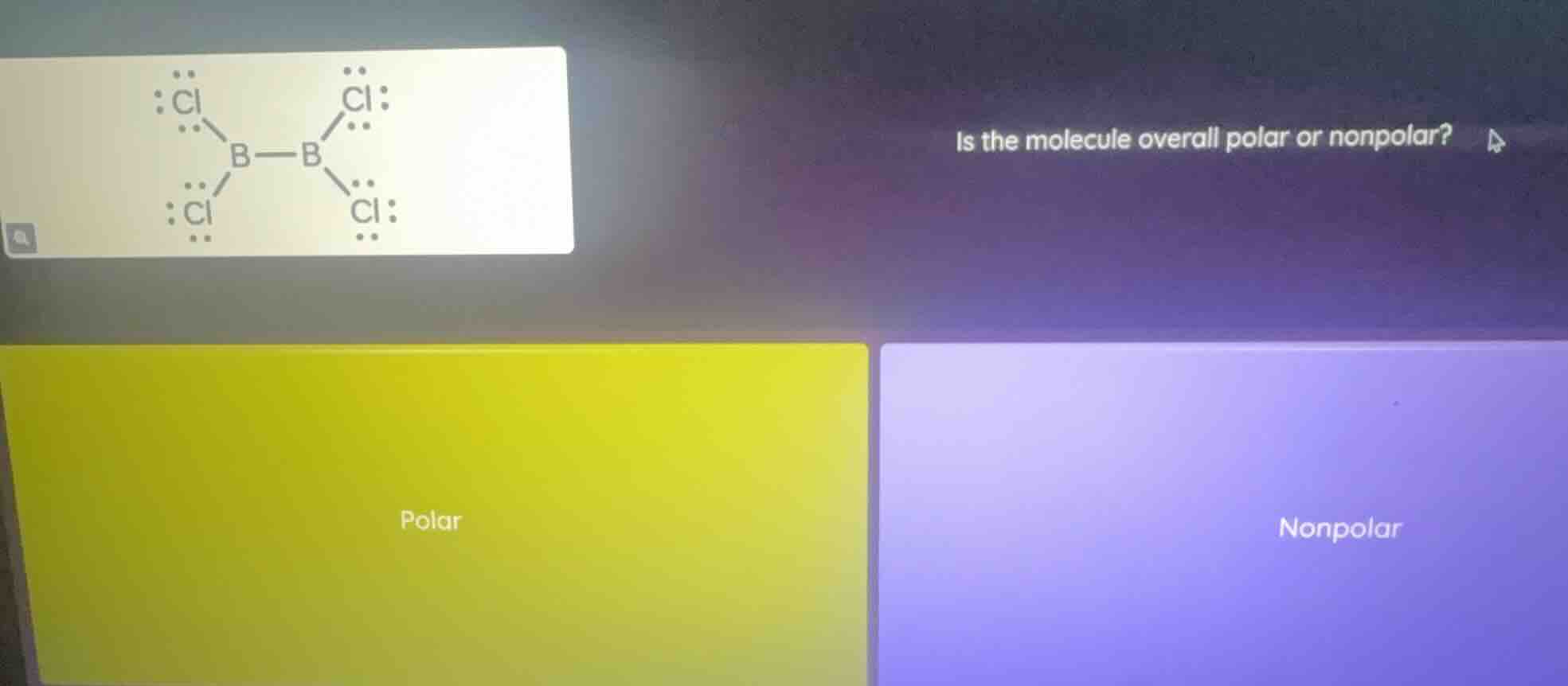
Question
is the molecule overall polar or nonpolar? polar nonpolar (with the molecular structure image of b - b bonded with four cl atoms)
To determine if the molecule (with structure \( \text{Cl}_2\text{B}_2\text{Cl}_2 \)) is polar or nonpolar, we analyze its symmetry. The molecule has a symmetrical arrangement of atoms (two B atoms bonded, each with two Cl atoms). Polar bonds (B - Cl) exist, but due to the symmetrical geometry, the bond dipoles cancel out. A molecule with symmetrical geometry and canceled bond dipoles is nonpolar.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Nonpolar