QUESTION IMAGE
Question
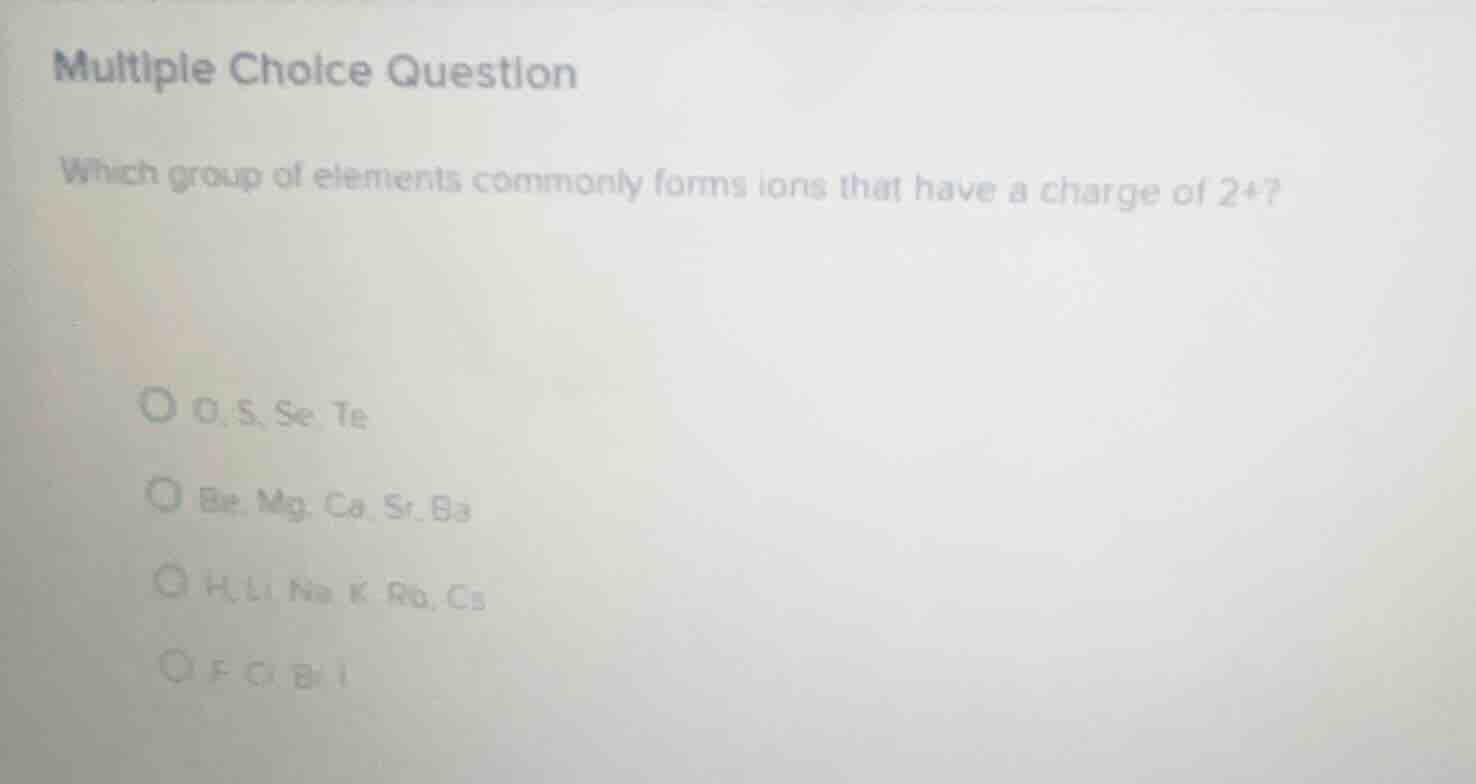
multiple choice question
which group of elements commonly forms ions that have a charge of 2+?
o, s, se, te
be, mg, ca, sr, ba
h, li, na, k, rb, cs
f, cl, br, i
Brief Explanations
Elements in Group 2 of the periodic table have 2 valence electrons. They tend to lose these 2 electrons to form stable cations with a +2 charge. The set Be, Mg, Ca, Sr, Ba are all Group 2 elements. The other options: O, S, Se, Te (Group 16) form -2 ions; H, Li, Na, K, Rb, Cs (Group 1) form +1 ions; F, Cl, Br, I (Group 17) form -1 ions.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Be, Mg, Ca, Sr, Ba