QUESTION IMAGE
Question
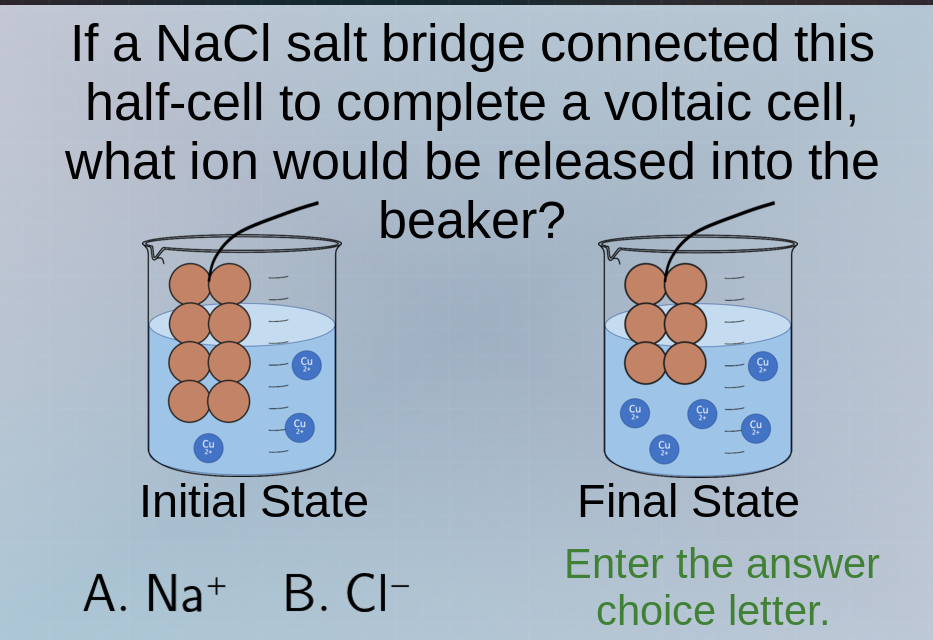
if a nacl salt bridge connected this half - cell to complete a voltaic cell, what ion would be released into the beaker? initial state final state a. na⁺ b. cl⁻ enter the answer choice letter.
In the initial state, the left beaker has fewer $\ce{Cu^{2+}}$ ions than the right. As the reaction proceeds (final state), the left beaker's $\ce{Cu^{2+}}$ concentration increases (more $\ce{Cu^{2+}}$ ions, likely from a reduction or oxidation process affecting charge). The salt bridge (NaCl) provides ions to balance charge. The left beaker is becoming more positive (more $\ce{Cu^{2+}}$), so anions (negative ions) from the salt bridge will move in to balance. $\ce{Cl^-}$ is the anion from NaCl, so $\ce{Cl^-}$ will be released into the left beaker (the one with the initial lower $\ce{Cu^{2+}}$) to neutralize the increasing positive charge.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. $\ce{Cl^-}$