QUESTION IMAGE
Question
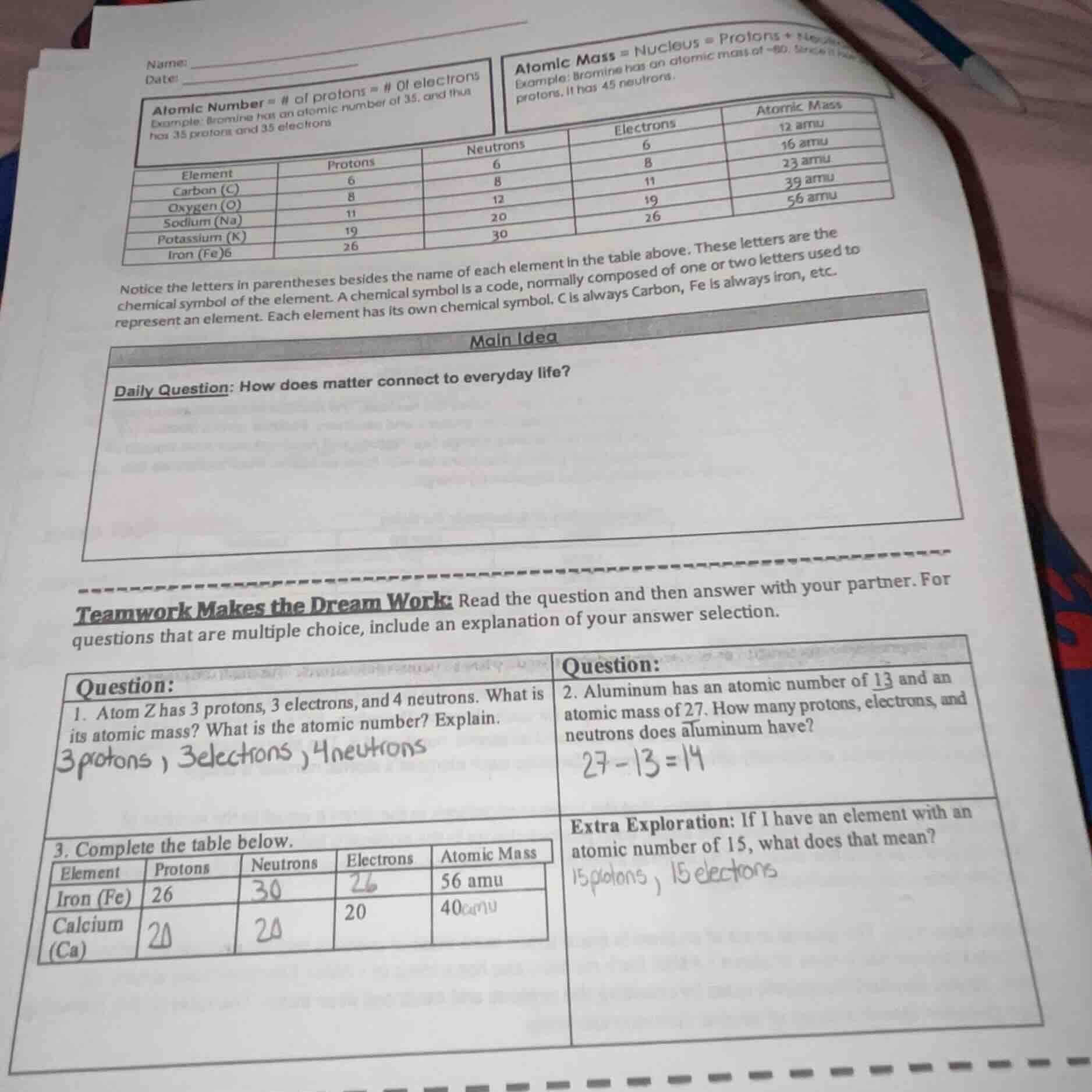
name:
date:
atomic number = # of protons = # of electrons
example: bromine has an atomic number of 35, and thus has 35 protons and 35 electrons
atomic mass = nucleus = protons +
example: bromine has an atomic mass of 80. since it has protons, it has 45 neutrons.
element\tprotons\tneutrons\telectrons\tatomic mass
carbon (c)\t6\t6\t6\t12 amu
oxygen (o)\t8\t8\t8\t16 amu
sodium (na)\t11\t12\t11\t23 amu
potassium (k)\t19\t20\t19\t39 amu
iron (fe)\t26\t30\t26\t56 amu
notice the letters in parentheses besides the name of each element in the table above. these letters are the chemical symbol of the element. a chemical symbol is a code, normally composed of one or two letters used to represent an element. each element has its own chemical symbol. c is always carbon, fe is always iron, etc.
main idea
daily question: how does matter connect to everyday life?
teamwork makes the dream work: read the question and then answer with your partner. for questions that are multiple choice, include an explanation of your answer selection.
question:
- atom z has 3 protons, 3 electrons, and 4 neutrons. what is its atomic mass? what is the atomic number? explain.
question:
- aluminum has an atomic number of 13 and an atomic mass of 27. how many protons, electrons, and neutrons does aluminum have?
- complete the table below.
element\tprotons\tneutrons\telectrons\tatomic mass
iron (fe)\t26\t\t\t56 amu
calcium (ca)\t\t\t20\t40 amu
extra exploration: if i have an element with an atomic number of 15, what does that mean?
Question 1:
Step1: Recall atomic mass formula
Atomic mass is the sum of protons and neutrons, i.e., \( \text{Atomic Mass} = \text{Protons} + \text{Neutrons} \). Atomic number equals the number of protons (and electrons in a neutral atom).
Step2: Calculate atomic mass for Atom Z
Protons = 3, Neutrons = 4. So atomic mass \( = 3 + 4 = 7 \) amu. Atomic number = number of protons = 3.
Step1: Use atomic number definition
Atomic number = number of protons = number of electrons (in neutral atom). So protons = 13, electrons = 13.
Step2: Calculate neutrons
Atomic mass = protons + neutrons, so neutrons = atomic mass - protons = \( 27 - 13 = 14 \).
Step1: Find neutrons for Iron
Atomic mass = protons + neutrons. Protons = 26, Atomic mass = 56. So neutrons \( = 56 - 26 = 30 \).
Step2: Find electrons for Iron
In neutral atom, electrons = protons = 26.
Calcium (Ca) row:
Step3: Find protons for Calcium
Electrons = 20 (neutral atom, so protons = electrons = 20).
Step4: Find neutrons for Calcium
Atomic mass = 40, protons = 20. So neutrons \( = 40 - 20 = 20 \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Atomic mass: 7 amu, Atomic number: 3. Explanation: Atomic mass is protons + neutrons (3 + 4 = 7), atomic number is number of protons (3).