QUESTION IMAGE
Question
name: _______________
date: _______________
teamwork makes the dream work: read the question and then answer with your partner. for questions that are multiple choice, include an explanation of your answer selection.
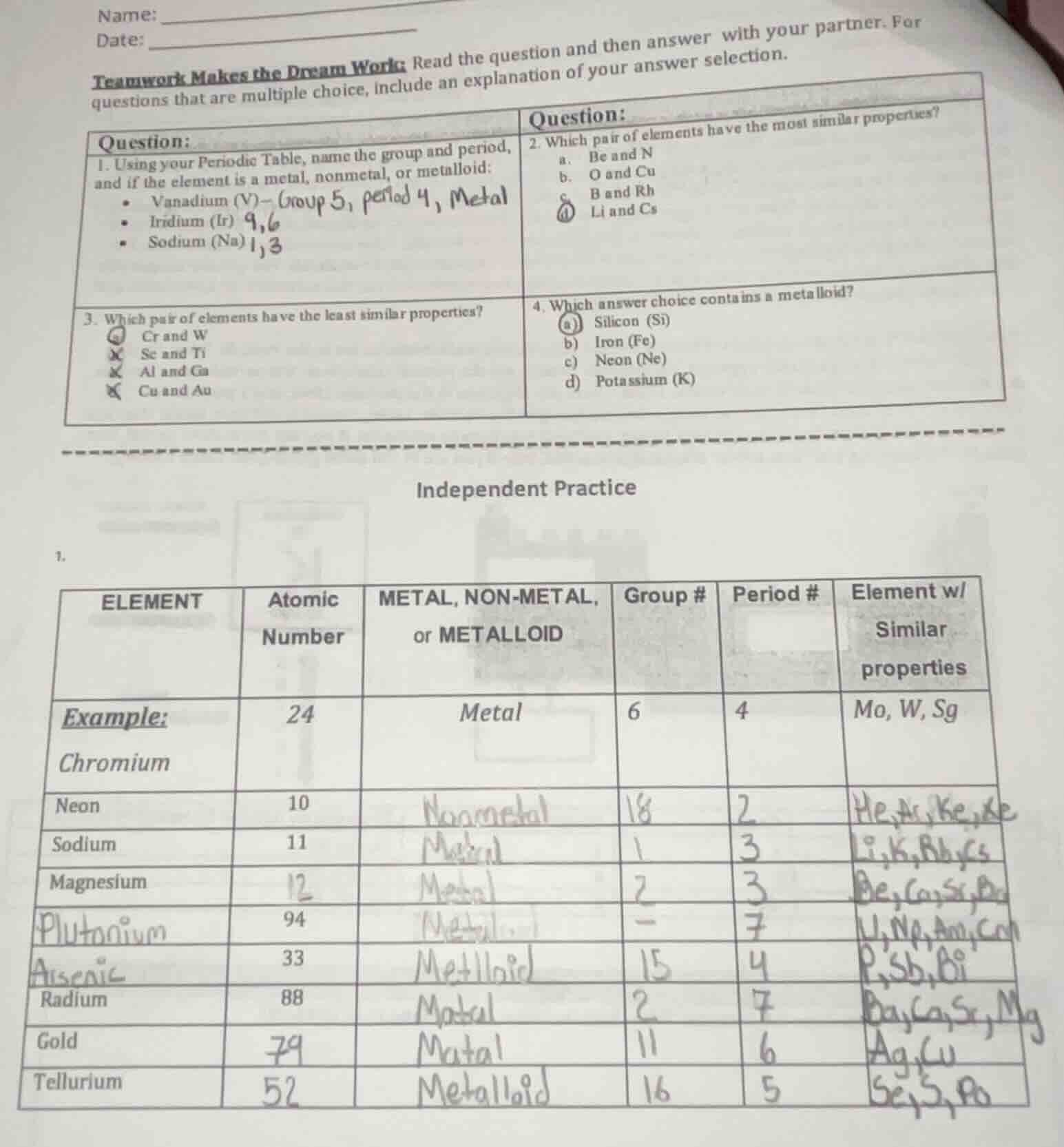
question:
- using your periodic table, name the group and period, and if the element is a metal, nonmetal, or metalloid:
- vanadium (v)
- iridium (ir)
- sodium (na)
question:
- which pair of elements have the most similar properties?
a. be and n
b. o and cu
c. b and rh
d. li and cs
- which pair of elements have the least similar properties?
a. cr and w
b. sc and ti
c. al and ga
d. cu and au
- which answer choice contains a metalloid?
a) silicon (si)
b) iron (fe)
c) neon (ne)
d) potassium (k)
independent practice
1.
| element | atomic number | metal, non - metal, or metalloid | group # | period # | element w/ similar properties |
|---|---|---|---|---|---|
| neon | 10 | ||||
| sodium | 11 | ||||
| magnesium | |||||
| 94 | |||||
| 33 | |||||
| radium | 88 | ||||
| gold | |||||
| tellurium |
Question 1 (Element Classification and Periodic Table Info)
Vanadium (V)
Step 1: Determine Group, Period, and Type
- Group: Vanadium is in Group 5 (transition metals, also known as Group VB in some numbering, but modern IUPAC Group 5).
- Period: Period 4 (since it has 4 electron shells).
- Type: Metal (all transition metals are metals).
Iridium (Ir)
Step 1: Determine Group, Period, and Type
- Group: Iridium is in Group 9 (IUPAC Group 9, part of the platinum group metals).
- Period: Period 6 (6 electron shells).
- Type: Metal (platinum group metals are metals).
Sodium (Na)
Step 1: Determine Group, Period, and Type
- Group: Group 1 (alkali metals).
- Period: Period 3 (3 electron shells).
- Type: Metal (alkali metals are metals).
Question 2 (Similar Properties)
Elements in the same group (vertical column) of the periodic table have similar properties because they have the same number of valence electrons.
- Option a: Be (Group 2) and N (Group 15) – different groups.
- Option b: O (Group 16) and Cu (Group 11) – different groups.
- Option c: B (Group 13) and Rh (Group 9) – different groups.
- Option d: Li (Group 1) and Cs (Group 1) – same group (alkali metals), so they have similar properties (e.g., both are highly reactive metals, form +1 ions, react vigorously with water).
Elements with the least similar properties are from different groups and different types (metal/non - metal/metalloid) or very different reactivity patterns.
- Cr (Group 6, metal) and W (Group 6, metal) – same group, similar properties.
- Sc (Group 3, metal) and Ti (Group 4, metal) – both transition metals, adjacent groups, some similarity.
- Al (Group 13, metal) and Cu (Group 11, metal) – different groups, but both metals.
- Cu (Group 11, metal) and Au (Group 11, metal) – same group, similar properties. Wait, there might be a mistake in the original markings. If we re - evaluate, Sc (Group 3, period 4) and Ti (Group 4, period 4) are adjacent, but Al (Group 13, period 3) and Cu (Group 11, period 4) are from different groups and periods, and have different properties (Al is a post - transition metal, Cu is a transition metal with different reactivity). But based on the original marked “X” on Al and Cu, the reasoning is that Al (Group 13, metal) and Cu (Group 11, metal) are in different groups with different valence electron configurations, leading to less similar properties compared to the others.
- Metalloids have properties intermediate between metals and non - metals.
- Option a: Silicon (Si) – metalloid (it has properties of both metals and non - metals, used in semiconductors).
- Option b: Iron (Fe) – metal.
- Option c: Neon (Ne) – non - metal (noble gas).
- Option d: Potassium (K) – metal (alkali metal).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
d. Li and Cs