QUESTION IMAGE
Question
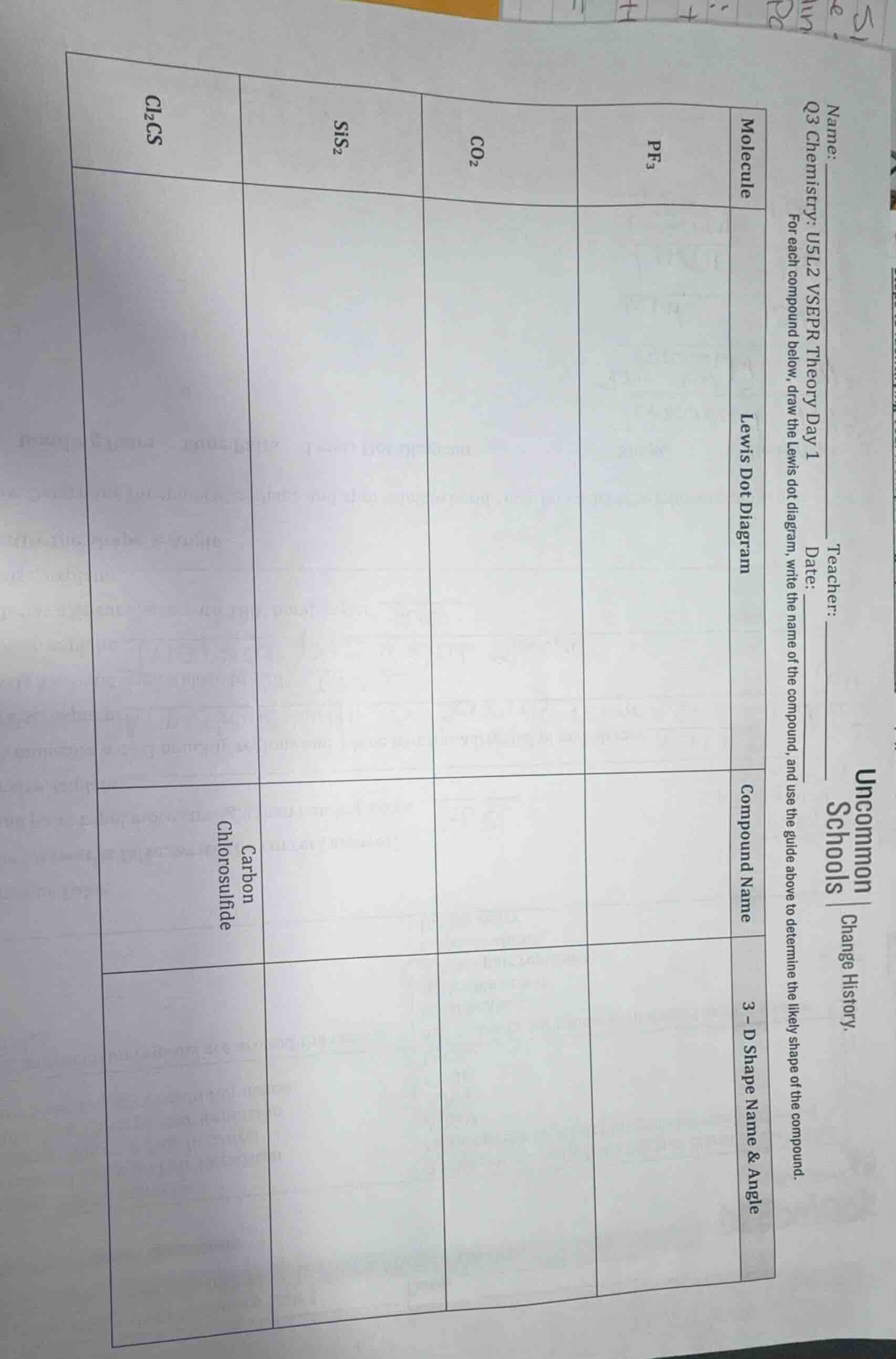
name:
teacher:
date:
q3 chemistry: u5l2 vsepr theory day 1
for each compound below, draw the lewis dot diagram, write the name of the compound, and use the guide above to determine the likely shape of the compound.
| molecule | lewis dot diagram | compound name | 3 - d shape name & angle |
|---|---|---|---|
| co₂ | |||
| sis₂ | |||
| cl₂cs | carbon chlorosulfide |
To solve this problem, we will analyze each molecule one by one, determining the Lewis Dot Diagram, Compound Name, and 3 - D Shape Name & Angle using VSEPR theory.
Molecule: \( \text{PF}_3 \)
Step 1: Lewis Dot Diagram
- Valence Electrons: P has 5 valence electrons, F has 7 valence electrons. For \( \text{PF}_3 \), total valence electrons \( = 5+3\times7 = 5 + 21=26 \).
- P is the central atom. It forms single bonds with 3 F atoms. Each single bond uses 2 electrons. So, 3 bonds use \( 3\times2 = 6 \) electrons. The remaining electrons \(=26 - 6=20\). These are distributed as lone pairs. P has 1 lone pair (\( 2 \) electrons) and each F has 3 lone pairs (\( 6 \) electrons per F, \( 3\times6 = 18 \) electrons for 3 F atoms). The Lewis structure is: P in the center, single - bonded to 3 F atoms, with one lone pair on P and three lone pairs on each F.
Step 2: Compound Name
The compound is named Phosphorus trifluoride.
Step 3: 3 - D Shape Name & Angle
- Electron - Pair Geometry: The central atom P has 4 electron - groups (3 bonding groups and 1 lone pair). According to VSEPR theory, the electron - pair geometry is tetrahedral.
- Molecular Geometry: Since there is 1 lone pair and 3 bonding pairs, the molecular geometry (3 - D shape) is trigonal pyramidal. The bond angle is approximately \( 107^{\circ} \) (due to the lone pair - bonding pair repulsion which is greater than bonding pair - bonding pair repulsion, compressing the bond angle from the ideal tetrahedral angle of \( 109.5^{\circ} \)).
Molecule: \( \text{CO}_2 \)
Step 1: Lewis Dot Diagram
- Valence Electrons: C has 4 valence electrons, O has 6 valence electrons. For \( \text{CO}_2 \), total valence electrons \(=4 + 2\times6=4 + 12 = 16\).
- C is the central atom. It forms double bonds with 2 O atoms. Each double bond uses 4 electrons. So, 2 double bonds use \( 2\times4 = 8 \) electrons. The remaining electrons \(=16 - 8 = 8\), which are distributed as 2 lone pairs on each O atom. The Lewis structure is: O = C = O, with two lone pairs on each O.
Step 2: Compound Name
The compound is named Carbon dioxide.
Step 3: 3 - D Shape Name & Angle
- Electron - Pair Geometry: The central atom C has 2 electron - groups (2 bonding groups, no lone pairs). According to VSEPR theory, the electron - pair geometry is linear.
- Molecular Geometry: Since there are no lone pairs, the molecular geometry (3 - D shape) is linear. The bond angle is \( 180^{\circ} \).
Molecule: \( \text{SiS}_2 \)
Step 1: Lewis Dot Diagram
- Valence Electrons: Si has 4 valence electrons, S has 6 valence electrons. For \( \text{SiS}_2 \), total valence electrons \(=4+2\times6 = 4 + 12=16\).
- Si is the central atom. It forms double bonds with 2 S atoms. Each double bond uses 4 electrons. So, 2 double bonds use \( 2\times4 = 8 \) electrons. The remaining electrons \(=16 - 8 = 8\), which are distributed as 2 lone pairs on each S atom. The Lewis structure is: S = Si = S, with two lone pairs on each S.
Step 2: Compound Name
The compound is named Silicon disulfide.
Step 3: 3 - D Shape Name & Angle
- Electron - Pair Geometry: The central atom Si has 2 electron - groups (2 bonding groups, no lone pairs). According to VSEPR theory, the electron - pair geometry is linear.
- Molecular Geometry: Since there are no lone pairs, the molecular geometry (3 - D shape) is linear. The bond angle is \( 180^{\circ} \).
Molecule: \( \text{Cl}_2\text{CS} \) (Carbon disulfide - like with Cl instead of one S? Wait, the correct formula - related naming: Le…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this problem, we will analyze each molecule one by one, determining the Lewis Dot Diagram, Compound Name, and 3 - D Shape Name & Angle using VSEPR theory.
Molecule: \( \text{PF}_3 \)
Step 1: Lewis Dot Diagram
- Valence Electrons: P has 5 valence electrons, F has 7 valence electrons. For \( \text{PF}_3 \), total valence electrons \( = 5+3\times7 = 5 + 21=26 \).
- P is the central atom. It forms single bonds with 3 F atoms. Each single bond uses 2 electrons. So, 3 bonds use \( 3\times2 = 6 \) electrons. The remaining electrons \(=26 - 6=20\). These are distributed as lone pairs. P has 1 lone pair (\( 2 \) electrons) and each F has 3 lone pairs (\( 6 \) electrons per F, \( 3\times6 = 18 \) electrons for 3 F atoms). The Lewis structure is: P in the center, single - bonded to 3 F atoms, with one lone pair on P and three lone pairs on each F.
Step 2: Compound Name
The compound is named Phosphorus trifluoride.
Step 3: 3 - D Shape Name & Angle
- Electron - Pair Geometry: The central atom P has 4 electron - groups (3 bonding groups and 1 lone pair). According to VSEPR theory, the electron - pair geometry is tetrahedral.
- Molecular Geometry: Since there is 1 lone pair and 3 bonding pairs, the molecular geometry (3 - D shape) is trigonal pyramidal. The bond angle is approximately \( 107^{\circ} \) (due to the lone pair - bonding pair repulsion which is greater than bonding pair - bonding pair repulsion, compressing the bond angle from the ideal tetrahedral angle of \( 109.5^{\circ} \)).
Molecule: \( \text{CO}_2 \)
Step 1: Lewis Dot Diagram
- Valence Electrons: C has 4 valence electrons, O has 6 valence electrons. For \( \text{CO}_2 \), total valence electrons \(=4 + 2\times6=4 + 12 = 16\).
- C is the central atom. It forms double bonds with 2 O atoms. Each double bond uses 4 electrons. So, 2 double bonds use \( 2\times4 = 8 \) electrons. The remaining electrons \(=16 - 8 = 8\), which are distributed as 2 lone pairs on each O atom. The Lewis structure is: O = C = O, with two lone pairs on each O.
Step 2: Compound Name
The compound is named Carbon dioxide.
Step 3: 3 - D Shape Name & Angle
- Electron - Pair Geometry: The central atom C has 2 electron - groups (2 bonding groups, no lone pairs). According to VSEPR theory, the electron - pair geometry is linear.
- Molecular Geometry: Since there are no lone pairs, the molecular geometry (3 - D shape) is linear. The bond angle is \( 180^{\circ} \).
Molecule: \( \text{SiS}_2 \)
Step 1: Lewis Dot Diagram
- Valence Electrons: Si has 4 valence electrons, S has 6 valence electrons. For \( \text{SiS}_2 \), total valence electrons \(=4+2\times6 = 4 + 12=16\).
- Si is the central atom. It forms double bonds with 2 S atoms. Each double bond uses 4 electrons. So, 2 double bonds use \( 2\times4 = 8 \) electrons. The remaining electrons \(=16 - 8 = 8\), which are distributed as 2 lone pairs on each S atom. The Lewis structure is: S = Si = S, with two lone pairs on each S.
Step 2: Compound Name
The compound is named Silicon disulfide.
Step 3: 3 - D Shape Name & Angle
- Electron - Pair Geometry: The central atom Si has 2 electron - groups (2 bonding groups, no lone pairs). According to VSEPR theory, the electron - pair geometry is linear.
- Molecular Geometry: Since there are no lone pairs, the molecular geometry (3 - D shape) is linear. The bond angle is \( 180^{\circ} \).
Molecule: \( \text{Cl}_2\text{CS} \) (Carbon disulfide - like with Cl instead of one S? Wait, the correct formula - related naming: Let's re - check. Wait, the given name is Carbon chlorosulfide. Let's assume the correct formula - based analysis.
Step 1: Lewis Dot Diagram (assuming C is central)
- Valence Electrons: C has 4, Cl has 7, S has 6. For \( \text{Cl}_2\text{CS} \), total valence electrons \(=4 + 2\times7+6=4 + 14 + 6=24\).
- C is central. It forms single bonds with 2 Cl atoms and a double bond with S. Single bonds: 2 Cl - C bonds (4 electrons), double bond C = S (4 electrons). Total bonding electrons \(=4 + 4=8\). Remaining electrons \(=24 - 8 = 16\). Cl has 3 lone pairs (6 electrons each, \( 2\times6 = 12 \) electrons), S has 2 lone pairs (4 electrons). So, Lewis structure: C in center, single - bonded to 2 Cl, double - bonded to S, with 3 lone pairs on each Cl and 2 lone pairs on S.
Step 2: Compound Name
As given, Carbon chlorosulfide.
Step 3: 3 - D Shape Name & Angle
- Electron - Pair Geometry: Central atom C has 3 electron - groups (2 single bonds, 1 double bond). Electron - pair geometry is trigonal planar.
- Molecular Geometry: Since there are no lone pairs on C, molecular geometry is trigonal planar. The bond angle is approximately \( 120^{\circ} \).
Summary Table
| Molecule | Lewis Dot Diagram | Compound Name | 3 - D Shape Name & Angle |
|---|---|---|---|
| \( \text{CO}_2 \) | O = C = O, 2 lone pairs on each O | Carbon dioxide | Linear, \( 180^{\circ} \) |
| \( \text{SiS}_2 \) | S = Si = S, 2 lone pairs on each S | Silicon disulfide | Linear, \( 180^{\circ} \) |
| \( \text{Cl}_2\text{CS} \) | C central, single - bonded to 2 Cl, double - bonded to S, 3 lone pairs on each Cl, 2 lone pairs on S | Carbon chlorosulfide | Trigonal planar, \( \approx120^{\circ} \) |
(Note: The Lewis dot diagrams can be represented more precisely with electron - dot notation, but the above description gives the key features. For the 3 - D shape, the electron - pair and molecular geometries are determined based on VSEPR theory which considers the number of bonding and non - bonding electron pairs around the central atom.)