QUESTION IMAGE
Question
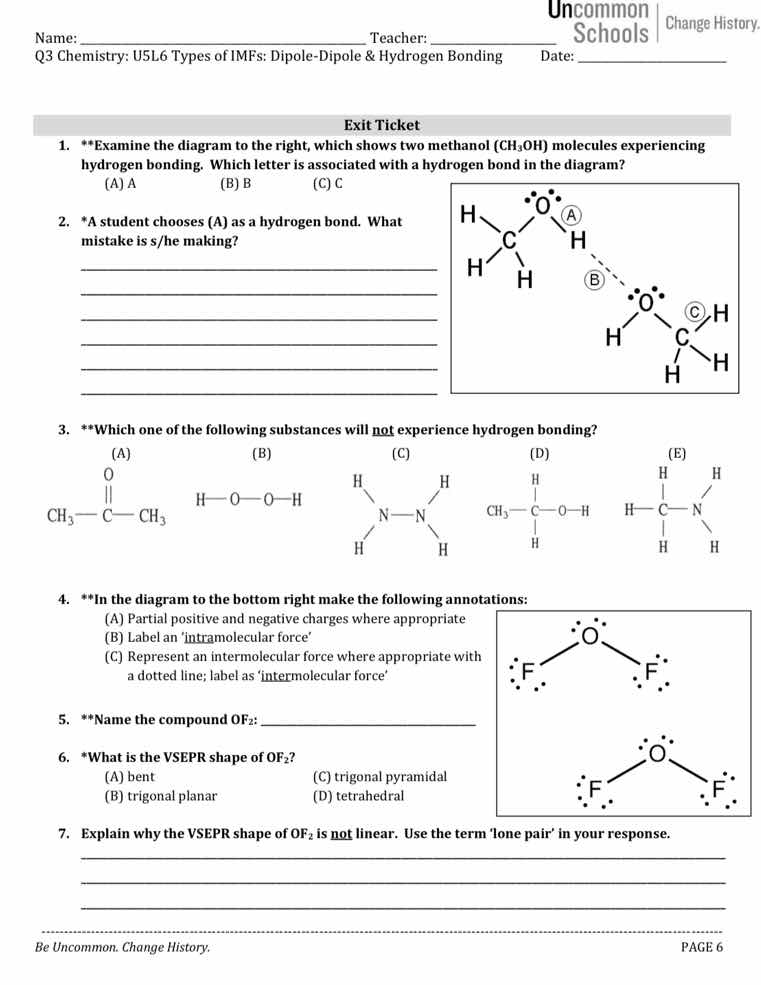
name: _________________________ teacher: _______________________q3 chemistry: u5l6 types of imfs: dipole-dipole & hydrogen bonding date: _______________________exit ticket1. *examine the diagram to the right, which shows two methanol (ch₃oh) molecules experiencing hydrogen bonding. which letter is associated with a hydrogen bond in the diagram?(a) a (b) b (c) c2. a student chooses (a) as a hydrogen bond. what mistake is s/he making?___________________________________________________________________________________________________________________________________________________________________________________________________________3. which one of the following substances will not experience hydrogen bonding?(a) $ce{ch3-c(=o)-ch3}$ (b) $ce{h-o-o-h}$ (c) $ce{h2n-nh2}$ (d) $ce{ch3-ch2-o-h}$ (e) $ce{h2n-ch2-h}$4. in the diagram to the bottom right make the following annotations:(a) partial positive and negative charges where appropriate(b) label an intramolecular force(c) represent an intermolecular force where appropriate with a dotted line; label as intermolecular force5. *name the compound of₂: _______________________6. what is the vsepr shape of of₂?(a) bent (b) trigonal planar (c) trigonal pyramidal (d) tetrahedral7. explain why the vsepr shape of of₂ is not linear. use the term lone pair in your response.________________________________________________________________________________________________________________________________________
Step1: Identify H-bond location
Hydrogen bonds are intermolecular attractions between a hydrogen bonded to N/O/F and a lone pair on another N/O/F. In the diagram, B is the dotted line between the H (bonded to O) of one methanol and the O of the other methanol.
Step2: Analyze mistake for (A)
(A) is a covalent bond between O and H within a single methanol molecule, not an intermolecular hydrogen bond.
Step3: Identify no H-bond substance
Hydrogen bonding requires H directly bonded to N/O/F. In option (A) $\text{CH}_3\text{COCH}_3$, all H are bonded to C, so no H-bonding occurs.
Step4: Annotate $\text{OF}_2$ diagram
(A) Partial charges: O is more electronegative than F, so O gets $\delta^-$, each F gets $\delta^+$.
(B) Intramolecular force: Label the single bonds between O and each F as 'intramolecular force'.
(C) Intermolecular force: Draw a dotted line between the $\delta^-$ O of one $\text{OF}_2$ and $\delta^+$ F of another $\text{OF}_2$, label as 'intermolecular force'.
Step5: Name $\text{OF}_2$
Use binary covalent naming: less electronegative element first, add -ide suffix with prefix for 2 F atoms.
Step6: Determine VSEPR shape of $\text{OF}_2$
$\text{OF}_2$ has 2 bonding pairs and 2 lone pairs on central O, so electron domain geometry is tetrahedral, molecular shape is bent.
Step7: Explain non-linear shape
Lone pairs exert greater repulsion than bonding pairs, pushing the bonding pairs closer together.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- (B)
- The student confused an intramolecular covalent O-H bond (A) with an intermolecular hydrogen bond; hydrogen bonds are intermolecular attractions between molecules, not bonds within a single molecule.
- (A) $\text{CH}_3\text{COCH}_3$
4.
(A) Mark $\delta^-$ on the central O, $\delta^+$ on each F atom in the $\text{OF}_2$ structure.
(B) Label the solid lines between O and each F as 'intramolecular force'.
(C) Draw a dotted line between the O of one $\text{OF}_2$ molecule and an F of a second $\text{OF}_2$ molecule, label the dotted line as 'intermolecular force'.
- Oxygen difluoride
- (A) bent
- The central oxygen in $\text{OF}_2$ has two lone pairs of electrons. Lone pairs exert stronger electrostatic repulsion than bonding electron pairs, which compresses the bond angle between the oxygen and fluorine atoms, resulting in a bent shape instead of a linear one.