QUESTION IMAGE
Question
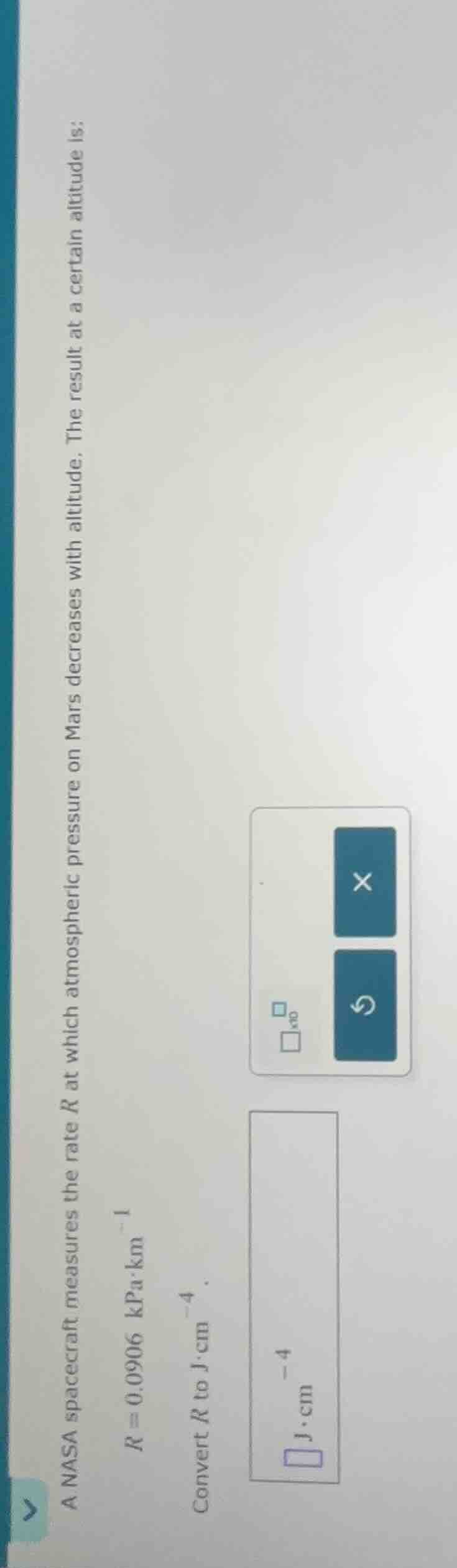
a nasa spacecraft measures the rate ( r ) at which atmospheric pressure on mars decreases with altitude. the result at a certain altitude is: ( r = 0.0906 space \text{kpa·km}^{-1} ) convert ( r ) to ( \text{j·cm}^{-4} ).
Step1: Recall unit conversion factors
$1\ \text{kPa} = 1000\ \text{Pa} = 1000\ \text{J·m}^{-3}$, $1\ \text{km} = 10^5\ \text{cm}$, $1\ \text{m}^3 = 10^6\ \text{cm}^3$
Step2: Convert kPa to J·cm⁻³
$0.0906\ \text{kPa} = 0.0906 \times 1000\ \text{J·m}^{-3} = 90.6\ \text{J·m}^{-3} = 90.6 \times 10^{-6}\ \text{J·cm}^{-3}$
Step3: Convert km⁻¹ to cm⁻¹
$\text{km}^{-1} = (10^5\ \text{cm})^{-1} = 10^{-5}\ \text{cm}^{-1}$
Step4: Combine units and calculate
$R = 90.6 \times 10^{-6}\ \text{J·cm}^{-3} \times 10^{-5}\ \text{cm}^{-1} = 9.06 \times 10^{-9}\ \text{J·cm}^{-4}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$9.06 \times 10^{-9}$