QUESTION IMAGE
Question
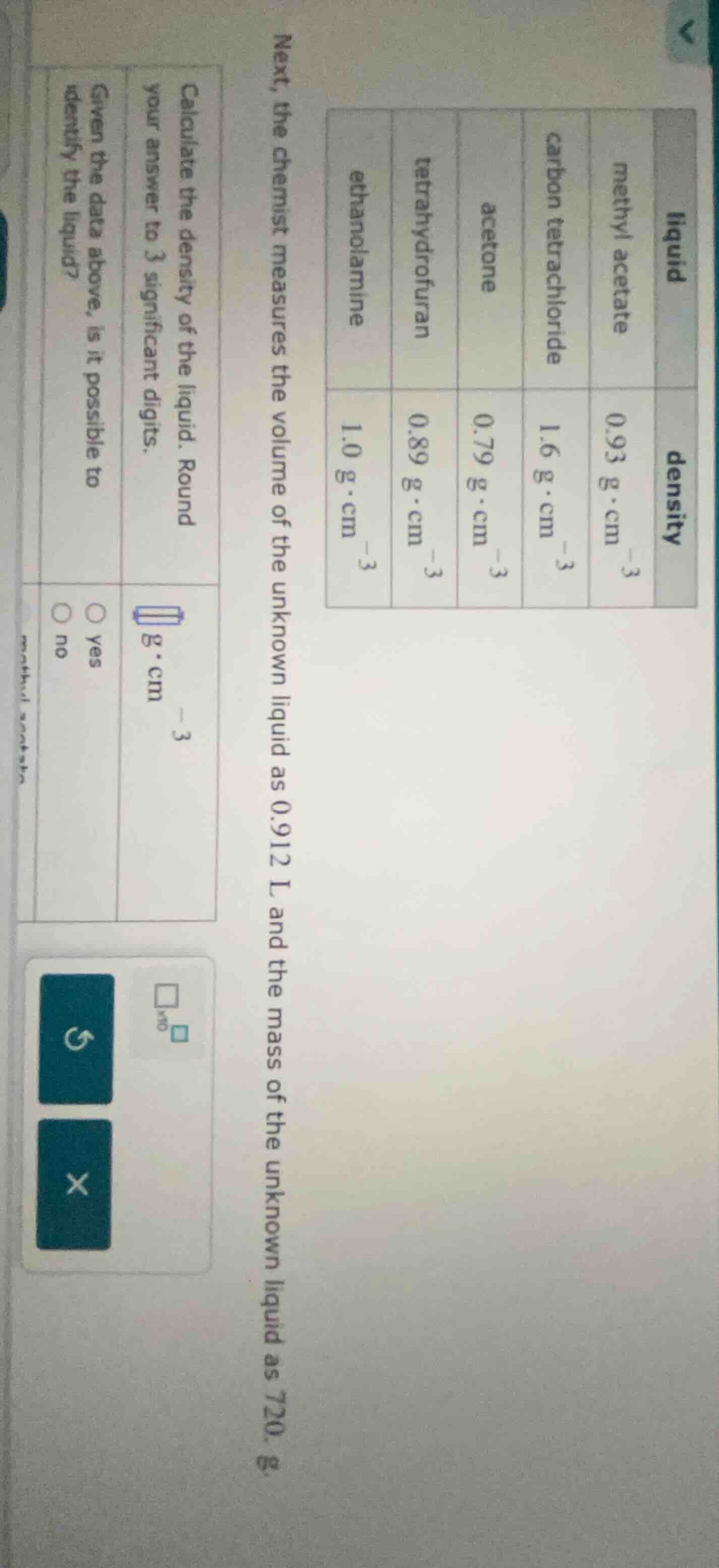
next, the chemist measures the volume of the unknown liquid as 0.912 l and the mass of the unknown liquid as 720. g.
calculate the density of the liquid. round your answer to 3 significant digits.
given the data above, is it possible to identify the liquid?
liquid | density (g·cm⁻³)
--- | ---
methyl acetate | 0.93carbon tetrachloride | 1.6
acetone | 0.79
tetrahydrofuran | 0.89
ethanolamine | 1.0
Step1: Convert volume to cm³
We know that \(1\space L = 1000\space cm^3\), so for a volume of \(0.912\space L\), we convert it to \(cm^3\) by multiplying by \(1000\).
\(0.912\space L\times1000 = 912\space cm^3\)
Step2: Use density formula (\(
ho=\frac{m}{V}\))
The density formula is \(
ho=\frac{m}{V}\), where \(m = 720\space g\) and \(V = 912\space cm^3\).
Substitute the values into the formula: \(
ho=\frac{720\space g}{912\space cm^3}\)
Step3: Calculate the density
\(\frac{720}{912}\approx0.789\space g\cdot cm^{-3}\) (rounded to 3 significant digits)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(0.789\space g\cdot cm^{-3}\)
For the second part (identifying the liquid):
Looking at the table, the density of acetone is \(0.79\space g\cdot cm^{-3}\), and our calculated density is \(0.789\space g\cdot cm^{-3}\) which is very close to acetone's density (considering significant figures and possible measurement errors). So the answer for "is it possible to identify the liquid" is yes, and the liquid is likely acetone. But since the first part was about calculating density, the density answer is \(0.789\space g\cdot cm^{-3}\)