QUESTION IMAGE
Question
- the next two parts are connected. answer each of the following.
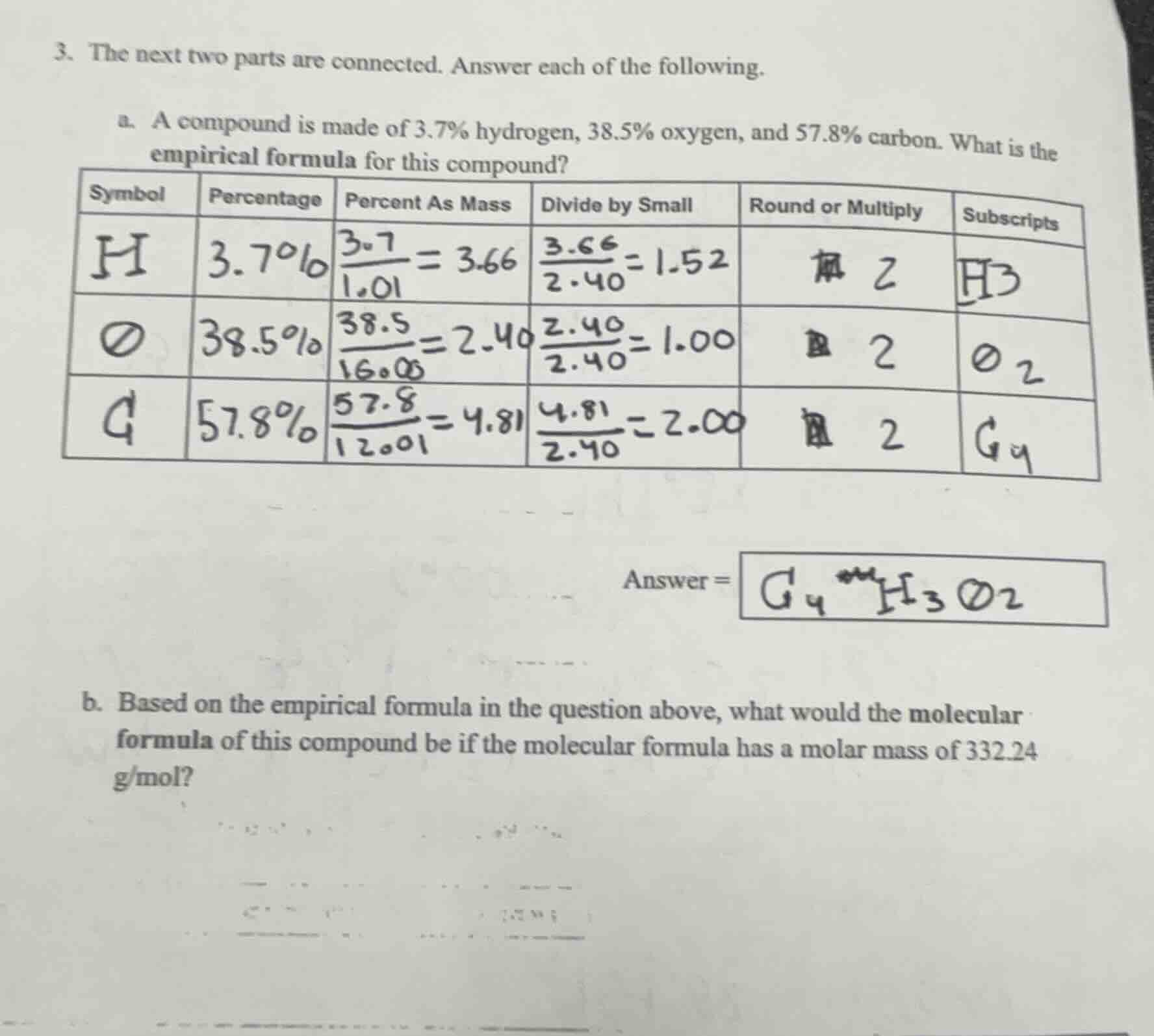
a. a compound is made of 3.7% hydrogen, 38.5% oxygen, and 57.8% carbon. what is the empirical formula for this compound?
| symbol | percentage | percent as mass | divide by small | round or multiply | subscripts |
| h | 3.7% | \\(\frac{3.7}{1.01}=3.66\\) | \\(\frac{3.66}{2.40}=1.52\\) | 2 | h3 |
| o | 38.5% | \\(\frac{38.5}{16.00}=2.40\\) | \\(\frac{2.40}{2.40}=1.00\\) | 2 | o2 |
| c | 57.8% | \\(\frac{57.8}{12.01}=4.81\\) | \\(\frac{4.81}{2.40}=2.00\\) | 2 | c4 |
answer = c₄h₃o₂
b. based on the empirical formula in the question above, what would the molecular formula of this compound be if the molecular formula has a molar mass of 332.24 g/mol?
Step1 (Part a): Verify mole calculation
For H: $\frac{3.7}{1.01} \approx 3.66$ mol
For O: $\frac{38.5}{16.00} = 2.406 \approx 2.40$ mol
For C: $\frac{57.8}{12.01} \approx 4.81$ mol
Step2 (Part a): Normalize by smallest mole
Divide each by 2.40:
H: $\frac{3.66}{2.40} = 1.52$
O: $\frac{2.40}{2.40} = 1.00$
C: $\frac{4.81}{2.40} \approx 2.00$
Step3 (Part a): Scale to whole numbers
Multiply all by 2 to eliminate decimals:
H: $1.52 \times 2 \approx 3$
O: $1.00 \times 2 = 2$
C: $2.00 \times 2 = 4$
Step4 (Part b): Calculate empirical formula mass
Empirical formula: $\text{C}_4\text{H}_3\text{O}_2$
Mass = $(4 \times 12.01) + (3 \times 1.01) + (2 \times 16.00)$
$= 48.04 + 3.03 + 32.00 = 83.07$ g/mol
Step5 (Part b): Find scaling factor
$n = \frac{\text{Molecular mass}}{\text{Empirical mass}} = \frac{332.24}{83.07} = 4$
Step6 (Part b): Get molecular formula
Multiply empirical subscripts by 4:
C: $4 \times 4 = 16$, H: $3 \times 4 = 12$, O: $2 \times 4 = 8$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a. $\text{C}_4\text{H}_3\text{O}_2$
b. $\text{C}_{16}\text{H}_{12}\text{O}_8$