QUESTION IMAGE
Question
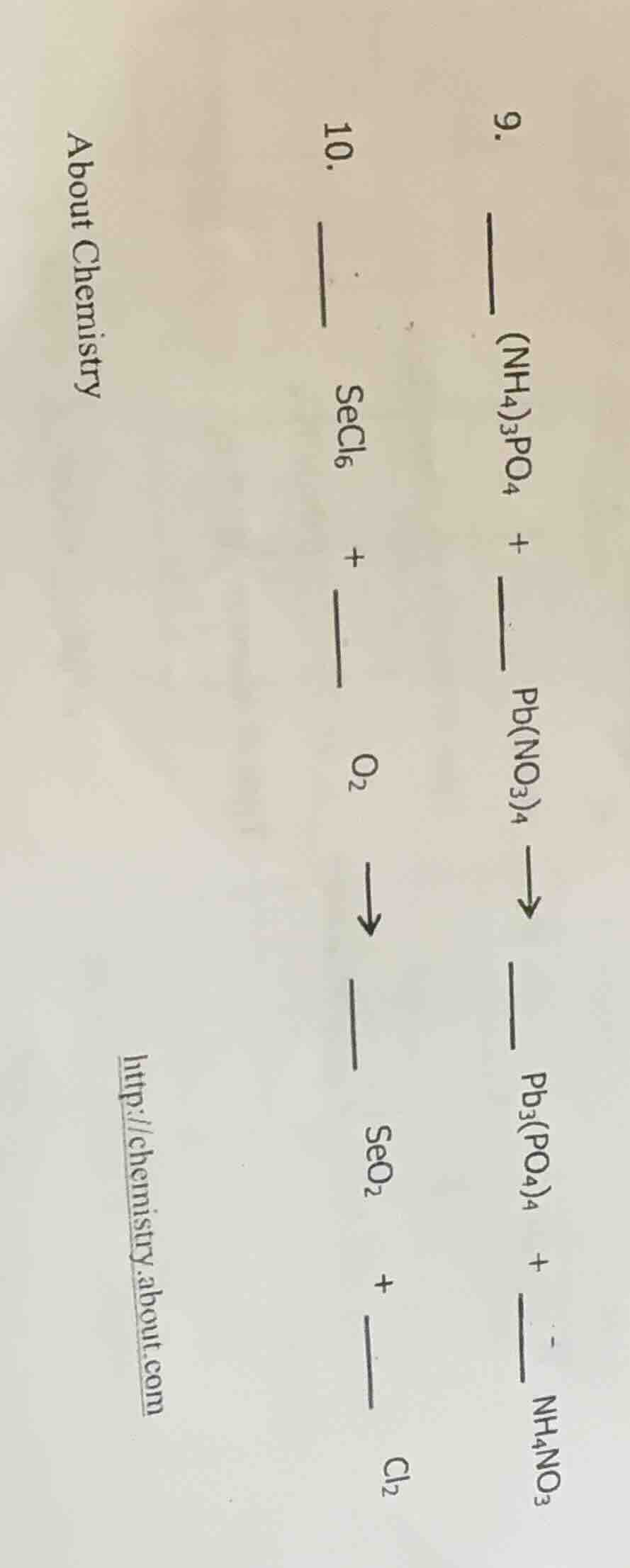
- _ (nh₄)₃po₄ + _ pb(no₃)₄ → _ pb₃(po₄)₄ + _ nh₄no₃
- _ secl₆ + _ o₂ → _ seo₂ + _ cl₂
Step1: Balance problem 9 (Pb atoms)
Count Pb: reactant has 1, product has 3. Multiply $\text{Pb(NO}_3\text{)}_4$ by 3.
Step2: Balance problem 9 (PO₄ groups)
Count $\text{PO}_4$: reactant has 1, product has 4. Multiply $\text{(NH}_4\text{)}_3\text{PO}_4$ by 4.
Step3: Balance problem 9 (NH₄⁺ groups)
Count $\text{NH}_4^+$: reactant has 12, product has 1. Multiply $\text{NH}_4\text{NO}_3$ by 12.
Step4: Verify problem 9 (NO₃⁻ groups)
Count $\text{NO}_3^-$: reactant has 12, product has 12. Equation is balanced.
Step5: Balance problem 10 (Se atoms)
Se is balanced (1 on each side).
Step6: Balance problem 10 (Cl atoms)
Count Cl: reactant has 6, product has 2. Multiply $\text{Cl}_2$ by 3.
Step7: Verify problem 10 (O atoms)
Count O: reactant has 2, product has 2. Equation is balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $4\text{(NH}_4\text{)}_3\text{PO}_4 + 3\text{Pb(NO}_3\text{)}_4
ightarrow \text{Pb}_3\text{(PO}_4\text{)}_4 + 12\text{NH}_4\text{NO}_3$
- $\text{SeCl}_6 + \text{O}_2
ightarrow \text{SeO}_2 + 3\text{Cl}_2$