QUESTION IMAGE
Question
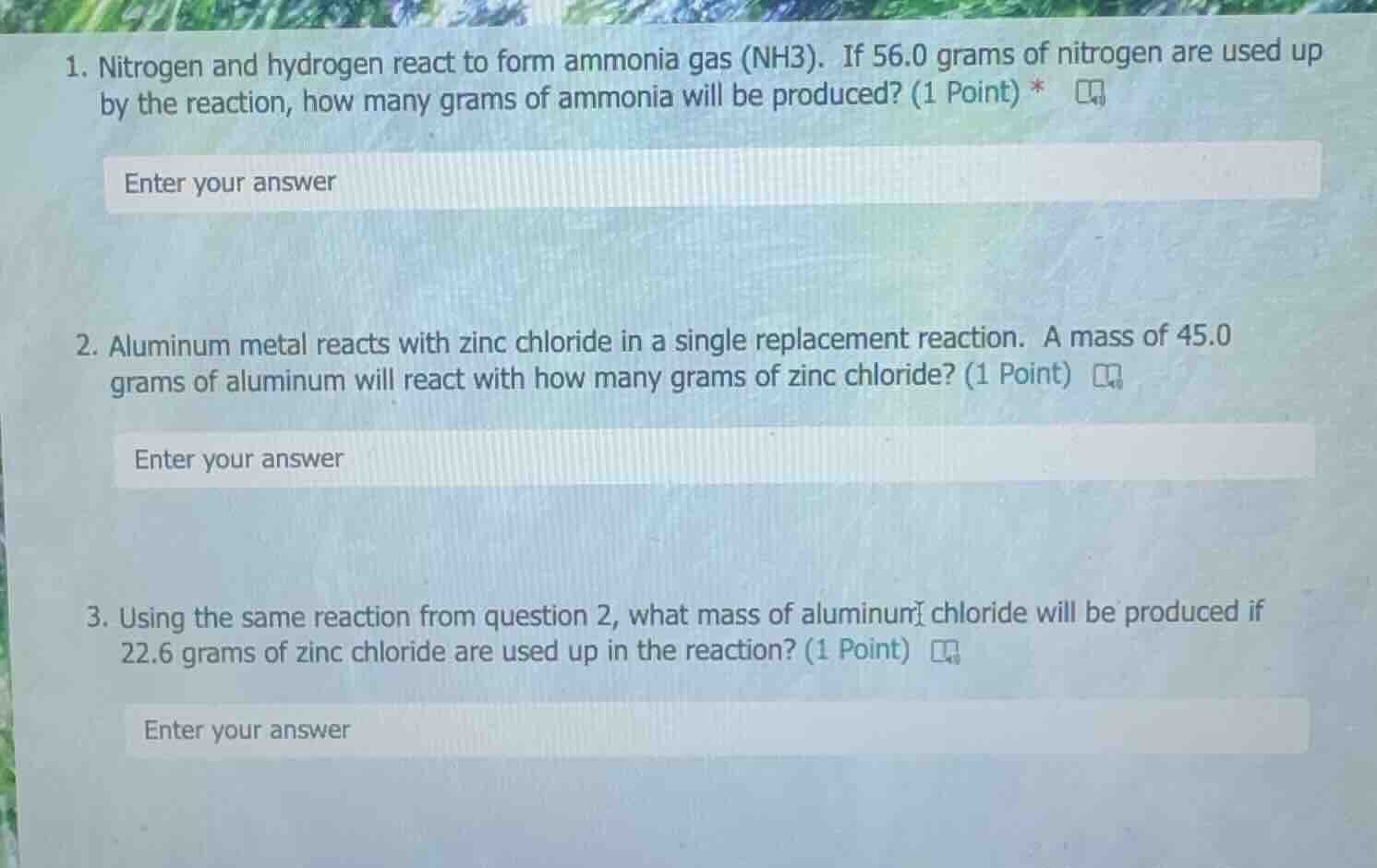
- nitrogen and hydrogen react to form ammonia gas (nh3). if 56.0 grams of nitrogen are used up by the reaction, how many grams of ammonia will be produced? (1 point) *
enter your answer
- aluminum metal reacts with zinc chloride in a single replacement reaction. a mass of 45.0 grams of aluminum will react with how many grams of zinc chloride? (1 point)
enter your answer
- using the same reaction from question 2, what mass of aluminum chloride will be produced if 22.6 grams of zinc chloride are used up in the reaction? (1 point)
enter your answer
Question 1
Step1: Write the balanced reaction
The reaction between nitrogen ($\ce{N2}$) and hydrogen ($\ce{H2}$) to form ammonia ($\ce{NH3}$) is given by the balanced equation: $\ce{N2 + 3H2 -> 2NH3}$
Step2: Calculate moles of $\ce{N2}$
Molar mass of $\ce{N2}$ is $2\times14 = 28\ \text{g/mol}$. Moles of $\ce{N2}$ used $=\frac{\text{mass}}{\text{molar mass}}=\frac{56.0\ \text{g}}{28\ \text{g/mol}} = 2\ \text{mol}$
Step3: Relate moles of $\ce{N2}$ to $\ce{NH3}$
From the balanced equation, 1 mole of $\ce{N2}$ produces 2 moles of $\ce{NH3}$. So, 2 moles of $\ce{N2}$ will produce $2\times2 = 4\ \text{mol}$ of $\ce{NH3}$
Step4: Calculate mass of $\ce{NH3}$
Molar mass of $\ce{NH3}$ is $14 + 3\times1 = 17\ \text{g/mol}$. Mass of $\ce{NH3}$ produced $=\text{moles}\times\text{molar mass}=4\ \text{mol}\times17\ \text{g/mol}=68.0\ \text{g}$
Step1: Write the balanced reaction
The single - replacement reaction between aluminum ($\ce{Al}$) and zinc chloride ($\ce{ZnCl2}$) is: $\ce{2Al + 3ZnCl2 -> 2AlCl3 + 3Zn}$
Step2: Calculate moles of $\ce{Al}$
Molar mass of $\ce{Al}$ is $27\ \text{g/mol}$. Moles of $\ce{Al}$ $=\frac{45.0\ \text{g}}{27\ \text{g/mol}}=\frac{5}{3}\ \text{mol}\approx1.6667\ \text{mol}$
Step3: Relate moles of $\ce{Al}$ to $\ce{ZnCl2}$
From the balanced equation, 2 moles of $\ce{Al}$ react with 3 moles of $\ce{ZnCl2}$. Let the moles of $\ce{ZnCl2}$ be $x$. Then, $\frac{2}{3}=\frac{\frac{5}{3}}{x}$, so $x=\frac{3\times\frac{5}{3}}{2}=\frac{5}{2} = 2.5\ \text{mol}$
Step4: Calculate mass of $\ce{ZnCl2}$
Molar mass of $\ce{ZnCl2}$ is $65.38+2\times35.45 = 65.38 + 70.9=136.28\ \text{g/mol}$. Mass of $\ce{ZnCl2}$ $=\text{moles}\times\text{molar mass}=2.5\ \text{mol}\times136.28\ \text{g/mol}=340.7\ \text{g}$ (approx)
Step1: Write the balanced reaction (same as question 2)
$\ce{2Al + 3ZnCl2 -> 2AlCl3 + 3Zn}$
Step2: Calculate moles of $\ce{ZnCl2}$
Molar mass of $\ce{ZnCl2}$ is $136.28\ \text{g/mol}$ (as calculated before). Moles of $\ce{ZnCl2}$ used $=\frac{22.6\ \text{g}}{136.28\ \text{g/mol}}\approx0.1658\ \text{mol}$
Step3: Relate moles of $\ce{ZnCl2}$ to $\ce{AlCl3}$
From the balanced equation, 3 moles of $\ce{ZnCl2}$ produce 2 moles of $\ce{AlCl3}$. Let moles of $\ce{AlCl3}$ be $x$. Then $\frac{3}{2}=\frac{0.1658\ \text{mol}}{x}$, so $x=\frac{2\times0.1658}{3}\approx0.1105\ \text{mol}$
Step4: Calculate mass of $\ce{AlCl3}$
Molar mass of $\ce{AlCl3}$ is $27+3\times35.45 = 27 + 106.35 = 133.35\ \text{g/mol}$. Mass of $\ce{AlCl3}$ produced $=0.1105\ \text{mol}\times133.35\ \text{g/mol}\approx14.74\ \text{g}$ (or using exact ratios)
Alternative way:
From the balanced equation, the mole ratio of $\ce{ZnCl2}$ to $\ce{AlCl3}$ is $3:2$.
Moles of $\ce{ZnCl2}=\frac{22.6\ \text{g}}{136.28\ \text{g/mol}}$
Moles of $\ce{AlCl3}=\frac{2}{3}\times\frac{22.6\ \text{g}}{136.28\ \text{g/mol}}$
Mass of $\ce{AlCl3}=\frac{2}{3}\times\frac{22.6\ \text{g}}{136.28\ \text{g/mol}}\times133.35\ \text{g/mol}$
$\frac{2\times22.6\times133.35}{3\times136.28}\approx\frac{2\times22.6\times133.35}{408.84}\approx\frac{6033.78}{408.84}\approx14.76\ \text{g}$ (approximate value, more precise calculation:
Molar mass of $\ce{ZnCl2}=65.38 + 2\times35.45=136.28\ \text{g/mol}$
Moles of $\ce{ZnCl2}=\frac{22.6}{136.28}=0.1658\ \text{mol}$
Moles of $\ce{AlCl3}=\frac{2}{3}\times0.1658 = 0.1105\ \text{mol}$
Molar mass of $\ce{AlCl3}=27+3\times35.45 = 133.35\ \text{g/mol}$
Mass of $\ce{AlCl3}=0.1105\times133.35 = 14.74\ \text{g}\approx14.7\ \text{g}$ (or 14.8 g depending on significant figures)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$68.0$ grams