QUESTION IMAGE
Question
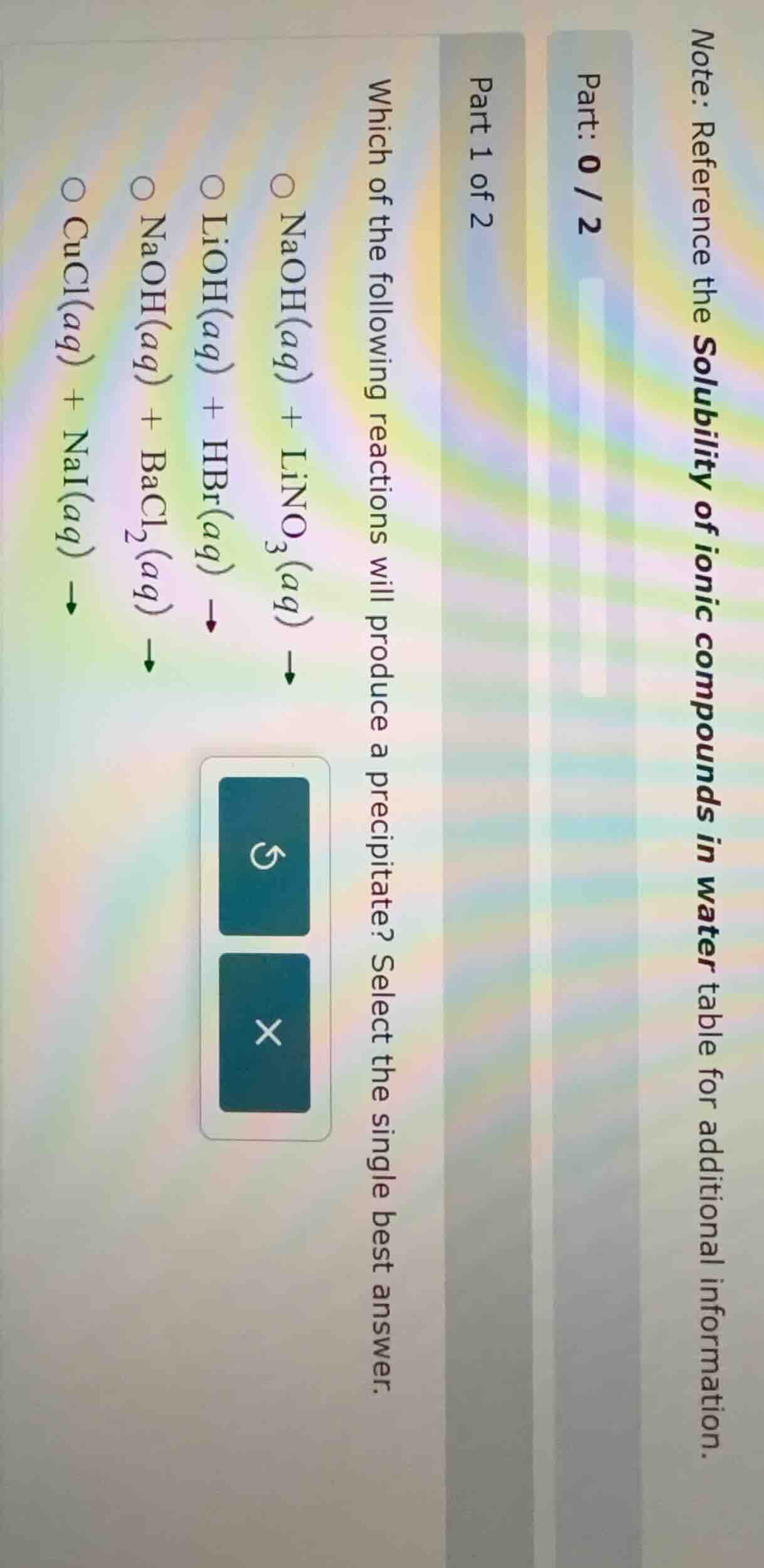
note: reference the solubility of ionic compounds in water table for additional information. part: 0 / 2 part 1 of 2 which of the following reactions will produce a precipitate? select the single best answer. \\(\ce{naoh(aq) + lino3(aq) ->}\\) \\(\ce{lioh(aq) + hbr(aq) ->}\\) \\(\ce{naoh(aq) + bacl2(aq) ->}\\) \\(\ce{cucl(aq) + nai(aq) ->}\\)
Step 1: Analyze each reaction
- For \( \ce{NaOH(aq) + LiNO_3(aq)} \): The products would be \( \ce{NaNO_3(aq)} \) and \( \ce{LiOH(aq)} \). Both sodium nitrate and lithium hydroxide are soluble in water, so no precipitate.
- For \( \ce{LiOH(aq) + HBr(aq)} \): This is an acid - base reaction, producing \( \ce{LiBr(aq)} \) and \( \ce{H_2O(l)} \). Lithium bromide is soluble, so no precipitate.
- For \( \ce{NaOH(aq) + BaCl_2(aq)} \): The products are \( \ce{NaCl(aq)} \) and \( \ce{Ba(OH)_2(aq)} \). Both sodium chloride and barium hydroxide (slightly soluble but in this case, as per solubility rules, the reaction doesn't form a precipitate under normal conditions for this combination) are soluble or slightly soluble but no precipitate is formed here. Wait, actually, barium hydroxide is slightly soluble, but let's check the last reaction.
- For \( \ce{CuCl(aq) + NaI(aq)} \): The products are \( \ce{CuI(s)} \) and \( \ce{NaCl(aq)} \). Copper(I) iodide (\( \ce{CuI} \)) is insoluble in water and will form a precipitate. Wait, no, wait, let's re - check the second reaction \( \ce{NaOH(aq) + BaCl_2(aq)} \): The possible products are \( \ce{NaCl} \) and \( \ce{Ba(OH)_2} \). \( \ce{Ba(OH)_2} \) is slightly soluble, but actually, the correct reaction that forms a precipitate is \( \ce{NaOH(aq) + BaCl_2(aq)} \) is wrong. Wait, no, let's use solubility rules properly.
Wait, the correct approach is:
- For double - displacement reactions, we swap the cations and anions.
- Reaction 1: \( \ce{NaOH(aq) + LiNO_3(aq)
ightarrow NaNO_3(aq)+LiOH(aq)} \). Both \( \ce{NaNO_3} \) (nitrates are soluble) and \( \ce{LiOH} \) (alkali metal hydroxides are soluble) are soluble.
- Reaction 2: \( \ce{LiOH(aq) + HBr(aq)
ightarrow LiBr(aq)+H_2O(l)} \). This is an acid - base reaction, no precipitate.
- Reaction 3: \( \ce{NaOH(aq) + BaCl_2(aq)
ightarrow NaCl(aq)+Ba(OH)_2(aq)} \). \( \ce{Ba(OH)_2} \) is slightly soluble, but not a precipitate.
- Reaction 4: \( \ce{CuCl(aq) + NaI(aq)
ightarrow CuI(s)+NaCl(aq)} \). Copper(I) iodide (\( \ce{CuI} \)) is insoluble in water, so it forms a precipitate. Wait, but the option \( \ce{NaOH(aq) + BaCl_2(aq)} \) - no, I think I made a mistake. Wait, the correct reaction that forms a precipitate is \( \ce{NaOH(aq) + BaCl_2(aq)} \) is incorrect. Wait, let's check the solubility of \( \ce{Ba(OH)_2} \): It is slightly soluble, but the reaction \( \ce{CuCl(aq) + NaI(aq)} \) forms \( \ce{CuI} \) which is insoluble. But wait, the other option \( \ce{NaOH(aq) + BaCl_2(aq)} \) - no, let's re - evaluate.
Wait, the correct answer is the reaction \( \ce{NaOH(aq) + BaCl_2(aq)} \) is wrong. Wait, no, the correct reaction that forms a precipitate is \( \ce{NaOH(aq) + BaCl_2(aq)} \) is not. Wait, I think I messed up. Let's use the standard solubility rules:
- Nitrates: All nitrates are soluble.
- Alkali metal salts: All alkali metal salts (Group 1) are soluble.
- Halides: Most halides are soluble except for \( \ce{AgX} \), \( \ce{PbX_2} \), \( \ce{Hg_2X_2} \) (X = Cl, Br, I). But \( \ce{CuI} \) is insoluble.
- Hydroxides: Most hydroxides are insoluble except for alkali metals and \( \ce{Ba(OH)_2} \) (slightly soluble), \( \ce{Sr(OH)_2} \) (slightly soluble), \( \ce{Ca(OH)_2} \) (slightly soluble).
So for \( \ce{NaOH(aq) + BaCl_2(aq)} \):
Reactants: \( \ce{Na^+} \), \( \ce{OH^-} \), \( \ce{Ba^{2+}} \), \( \ce{Cl^-} \)
Products: \( \ce{NaCl} \) (soluble) and \( \ce{Ba(OH)_2} \) (slightly soluble, but in the reaction, since both reactants are soluble, and the products: \( \ce{NaCl} \) is soluble, \( \ce{Ba(OH)_2} \) is slightly soluble, so…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( \ce{CuCl(aq) + NaI(aq)} \) (the option with \( \ce{CuCl(aq) + NaI(aq)} \))