QUESTION IMAGE
Question
do now 20 (module 7 - lesson 4)
3 of 4 01:38 / 05:00
possible points: 25
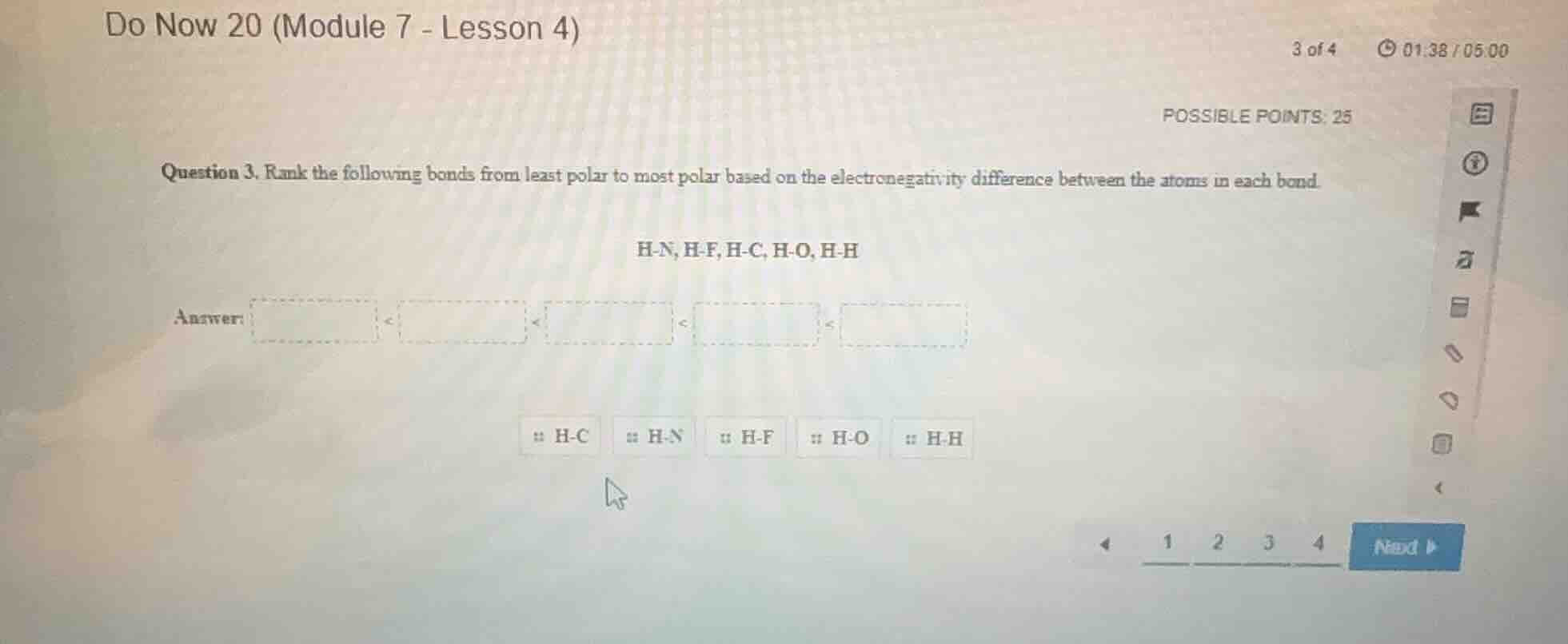
question 3. rank the following bonds from least polar to most polar based on the electronegativity difference between the atoms in each bond.
h-n, h-f, h-c, h-o, h-h
answer:
< < < <
h-c h-n h-f h-o h-h
Step1: Get electronegativity values
Electronegativity (Pauling scale): H=2.20, C=2.55, N=3.04, O=3.44, F=3.98
Step2: Calculate ΔEN for each bond
- H-H: $|2.20 - 2.20| = 0$
- H-C: $|2.20 - 2.55| = 0.35$
- H-N: $|2.20 - 3.04| = 0.84$
- H-O: $|2.20 - 3.44| = 1.24$
- H-F: $|2.20 - 3.98| = 1.78$
Step3: Rank by ΔEN (low to high)
Lower ΔEN = less polar; higher ΔEN = more polar.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
H-H < H-C < H-N < H-O < H-F