QUESTION IMAGE
Question
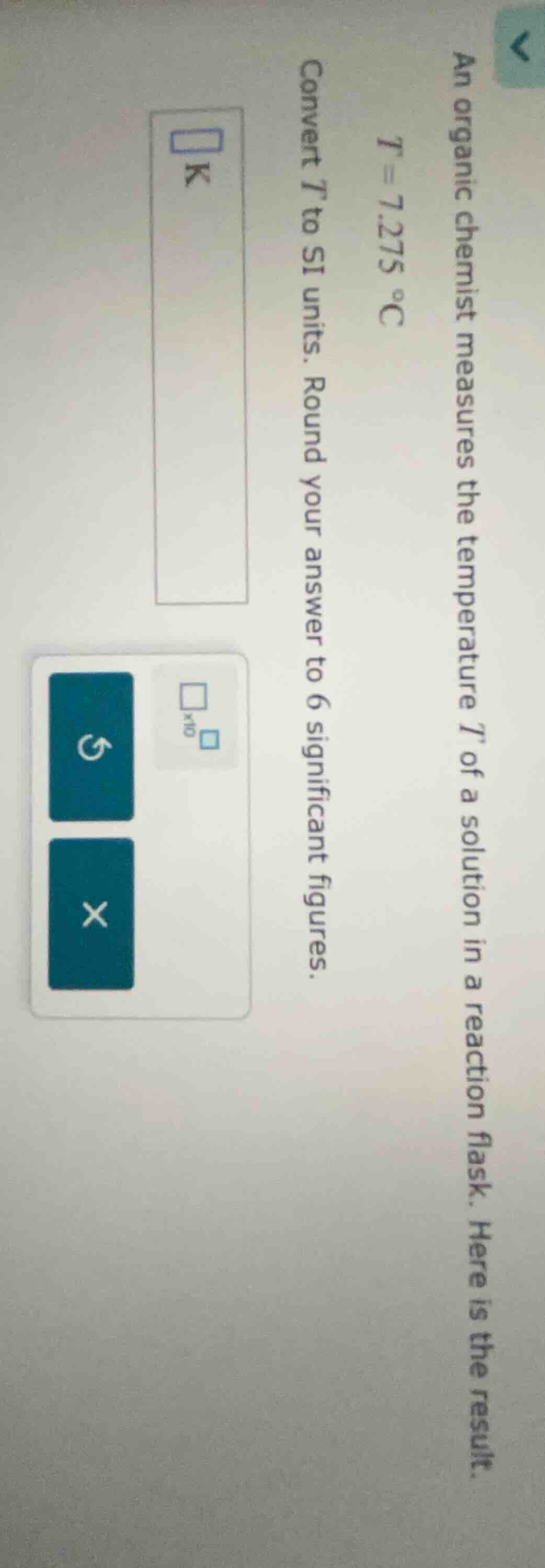
an organic chemist measures the temperature t of a solution in a reaction flask. here is the result. t = 7.275 °c convert t to si units. round your answer to 6 significant figures.
Step1: Recall the conversion formula from Celsius to Kelvin
The formula to convert temperature from degrees Celsius ($^\circ$C) to Kelvin (K) is $T(\text{K}) = T(^\circ\text{C}) + 273.15$.
Step2: Substitute the given value into the formula
We are given $T = 7.275^\circ\text{C}$. Substituting this into the formula, we get $T(\text{K}) = 7.275 + 273.15$.
Step3: Perform the addition
Calculating the sum: $7.275 + 273.15 = 280.425$.
Step4: Round to 6 significant figures
The number 280.425 already has 6 significant figures (the digits 2, 8, 0, 4, 2, 5; note that the zero here is significant as it is between non - zero digits and the number is written in a way that shows precision).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
280.425