QUESTION IMAGE
Question
organic chemistry
maxwell
presented by macmillan learning
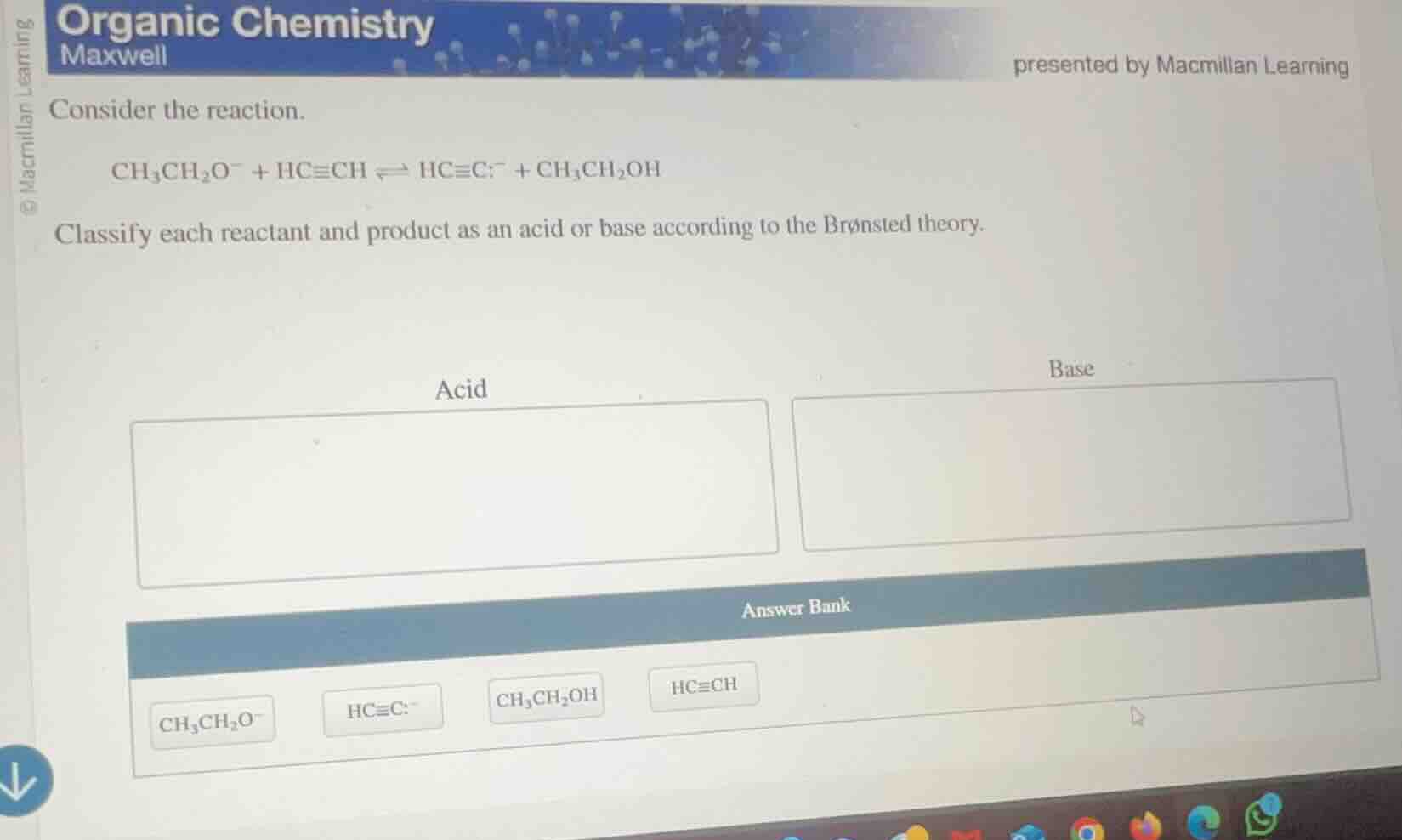
consider the reaction.
\ce{ch3ch2o- + hc#ch <=> hc#c- + ch3ch2oh}
classify each reactant and product as an acid or base according to the brønsted theory.
acid
base
answer bank
\ce{ch3ch2o-}
\ce{hc#c-}
\ce{ch3ch2oh}
\ce{hc#ch}
Brief Explanations
Per Brønsted theory, acids donate H⁺, bases accept H⁺.
- $\text{HC≡CH}$ donates an H⁺ to form $\text{HC≡C}^-$, so it is an acid.
- $\text{CH}_3\text{CH}_2\text{OH}$ is the product formed when $\text{CH}_3\text{CH}_2\text{O}^-$ accepts an H⁺, so it is also an acid (conjugate acid).
- $\text{CH}_3\text{CH}_2\text{O}^-$ accepts an H⁺ to form $\text{CH}_3\text{CH}_2\text{OH}$, so it is a base.
- $\text{HC≡C}^-$ is the product formed when $\text{HC≡CH}$ donates an H⁺, so it is also a base (conjugate base).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Acid:
$\text{HC≡CH}$, $\text{CH}_3\text{CH}_2\text{OH}$
Base:
$\text{CH}_3\text{CH}_2\text{O}^-$, $\text{HC≡C}^-$