QUESTION IMAGE
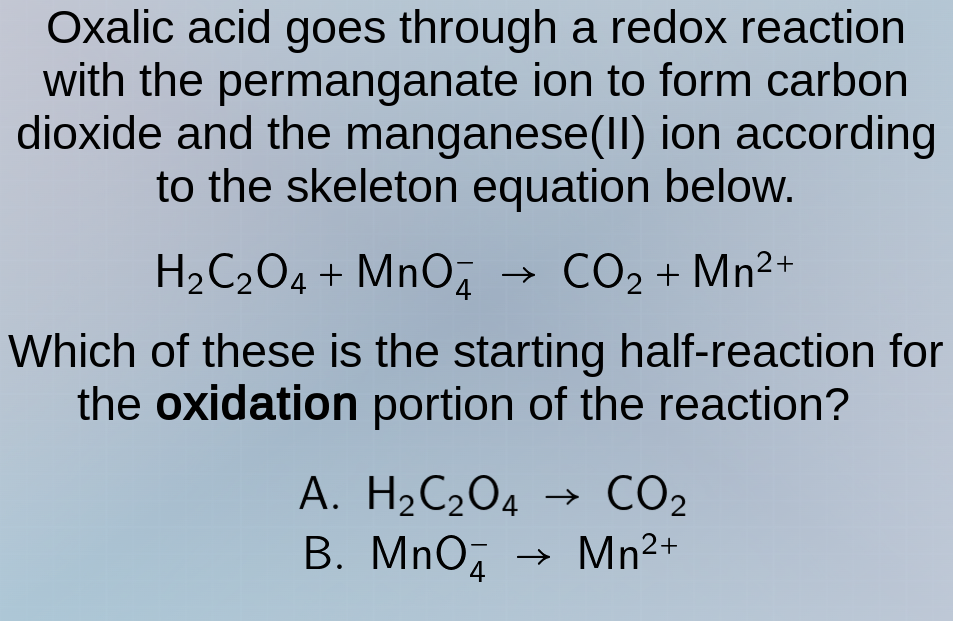
Question
oxalic acid goes through a redox reaction with the permanganate ion to form carbon dioxide and the manganese(ii) ion according to the skeleton equation below.
h₂c₂o₄ + mno₄⁻ → co₂ + mn²⁺
which of these is the starting half - reaction for the oxidation portion of the reaction?
a. h₂c₂o₄ → co₂
b. mno₄⁻ → mn²⁺
To determine the oxidation half - reaction, we look at the change in oxidation states. In \(H_2C_2O_4\), carbon has an oxidation state of +3. In \(CO_2\), carbon has an oxidation state of +4. An increase in oxidation state means oxidation. For \(MnO_4^-\), Mn has an oxidation state of +7, and in \(Mn^{2+}\), it has an oxidation state of +2, which is a decrease (reduction). So the oxidation half - reaction starts with the species where the element is oxidized, which is \(H_2C_2O_4\) being oxidized to \(CO_2\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. \( \ce{H_2C_2O_4
ightarrow CO_2} \)